Preparation of a thermo-sensitive gel uterine perfusate and drug concentration monitoring
-
摘要:目的
研制一种多西环素温敏型原位凝胶子宫灌注液,并对其进行质量评价及子宫内药物浓度的监测。
方法以泊洛沙姆407(P407)和泊洛沙姆188(P188)为主要温敏材料,采用“冷法”工艺制备原位凝胶。采用单因素分析和正交试验对处方进行筛选与优化,通过无膜溶出模型考察制剂体外溶蚀和释药情况,并在高温、高湿、强光等条件下对研制的温敏凝胶稳定性进行考察。选用5头二元杂交母猪对制剂进行子宫内药物浓度的监测。
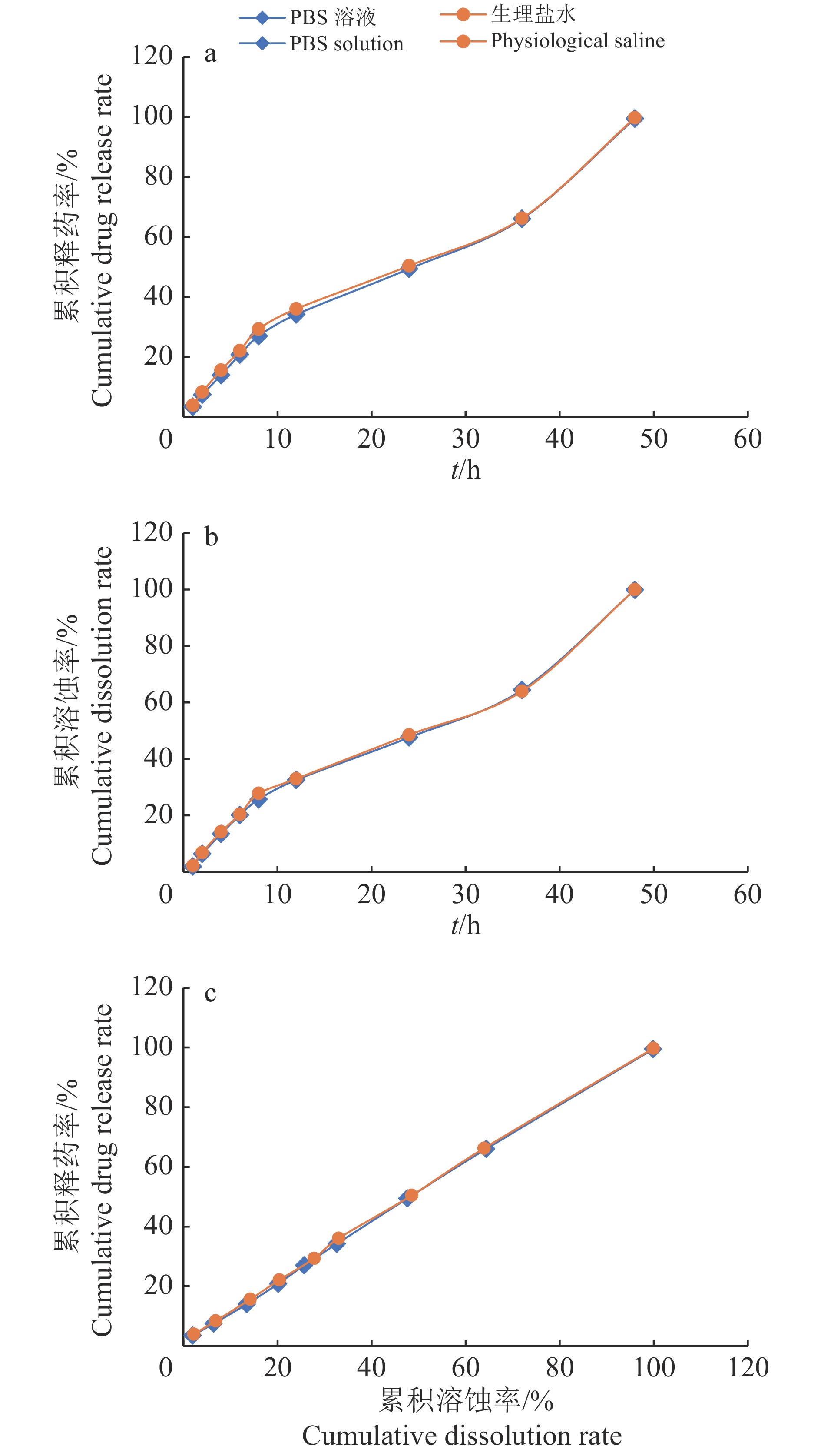
结果最终优化处方为50 g制剂含多西环素1 g、P407 10 g、P188 2.5 g、氯化镁2.3 g和亚硫酸氢钠0.1 g,用乙醇胺调pH至4.0±0.2,余量为超纯水。制剂体外溶蚀及释药时间长达48 h,累积溶蚀率和释药率均达100%。制剂稳定性符合兽用化学药物稳定性研究技术指导原则的要求,在第2次给药96 h后母猪子宫内药物浓度仍能保持在68.54 μg/mL。
结论多西环素温敏型原位凝胶子宫灌注液制备方法简单,性质稳定,猪子宫灌注给药后局部药物浓度长时间维持在较高水平,具有缓释效果,有望为临床治疗母猪子宫内膜炎开创新途径。
Abstract:ObjectiveTo prepare a doxycycline thermo-sensitive in situ gel uterine perfusate, evaluate its quality, and monitor drug concentration in sow uterus.
MethodPoloxamer 407 (P407) and poloxamer 188 (P188) were used as the main thermo-sensitive materials. The in situ gel was prepared by “cold” process. The prescription was screened and optimized by single factor analysis and orthogonal experiment. The in vitro dissolution and drug release of the preparation were measured by membrane free dissolution model. The stability of the developed thermo-sensitive gel was investigated under the conditions of high temperature, high humidity and strong light. Five binary hybrid sows were selected to monitor drug concentration in sow uterus.
ResultThe final optimized prescription was doxycycline 1 g, P407 10 g, P188 2.5 g, magnesium chloride 2.3 g and sodium bisulfite 0.1 g in per 50 g preparation, pH was adjusted to 4.0±0.2 with ethanolamine, and the residue was ultrapure water. The in vitro dissolution and drug release time of the preparation lasted for 48 h, the cumulative dissolution rate and drug release rate were up to 100%. The preparation stability was complied with technical guidelines for the stability of veterinary chemical drugs. The drug concentration remained at 68.54 μg/mL after 96 h of the second administration.
ConclusionThe doxycycline thermo-sensitive in situ gel uterine perfusate is a simple preparation method and shows good stability. The local concentration of the drug in sow uterus maintains at a high level for a long time. The preparation has a sustained release effect and is expected to be an innovative way to treat sow endometritis.
-
Keywords:
- doxycycline /
- thermo-sensitive in situ gel /
- poloxamer solution /
- sow endometritis /
- slow release
-
-
表 1 正交试验设计和结果1)
Table 1 Design and result of orthogonal experiment
试验号
Test number因素 Factor 多西环素含量变化/%
Doxycycline content change泊洛沙姆溶液
Poloxamer solution络合物摩尔比
Mole ratio
of complexw(亚硫酸氢钠)/%
Sodium bisulfite
contentpH w(P407)/% w(P188)/% 1 18 3 5∶1 0.10 pH0 1.83 2 18 3 3∶1 0.15 3.0 2.86 3 18 3 1∶1 0.20 4.0 1.50 4 20 5 5∶1 0.15 4.0 1.88 5 20 5 3∶1 0.20 pH0 1.82 6 20 5 1∶1 0.10 3.0 3.13 7 20 6 5∶1 0.20 3.0 1.03 8 20 6 3∶1 0.10 4.0 1.63 9 20 6 1∶1 0.15 pH0 4.33 平均值1/% Mean 1 2.063 1.580 2.197 2.660 平均值2/% Mean 2 2.277 2.103 3.023 2.340 平均值3/% Mean 3 2.330 2.987 1.450 1.670 极差 Range 0.267 1.407 1.573 0.990 1) 络合物摩尔比表示金属络合剂氧化镁与多西环素的摩尔比例;pH0表示初始pH;多西环素含量变化指制备试剂后第0天和第7天多西环素含量的差值;平均值1、2、3分别代表了4个考察因素为1、2、3水平时多西环素含量变化的平均值
1) Mole ratio of complex indicates mole ratio of metal complexing agent magnesium chloride to doxycycline; pH0 indicates initial pH; Doxycycline content change means doxycycline content difference between zero and seven days after preparing reagent; Mean 1, 2, 3 respectively indicate means of doxycycline content change of 1, 2, 3 levels of four factors表 2 多西环素温敏型原位凝胶子宫灌注液加速试验结果
Table 2 Results of acceleration test of doxycycline thermo-sensitive in situ gel uterine perfusate
批次
Batcht/d 性状
CharacterpH θ胶凝/℃
Gelling temperaturew(多西环素)/%
Doxycycline content1 0 淡黄色半固体凝胶
Pale yellow semi-solid gel4.04 29.9 99.91 30 4.05 30.3 98.90 60 4.02 30.0 99.14 90 4.02 30.1 99.86 180 4.03 30.2 99.74 2 0 淡黄色半固体凝胶
Pale yellow semi-solid gel4.05 30.1 99.84 30 4.01 30.2 98.83 60 4.03 30.0 99.12 90 4.02 29.8 99.83 180 4.02 30.2 98.67 3 0 淡黄色半固体凝胶
Pale yellow semi-solid gel4.02 30.4 99.10 30 4.03 30.1 98.63 60 4.01 30.3 98.39 90 4.01 30.3 99.27 180 4.02 30.1 98.58 表 3 多西环素温敏型原位凝胶子宫灌注液长期稳定性试验结果
Table 3 Results of long-term stability test of doxycycline thermo-sensitive in situ gel uterine perfusate
批次
Batcht/d 性状
CharacterpH θ胶凝/℃
Gelling temperaturew(多西环素)/%
Doxycycline content1 0 淡黄色澄清溶液
Pale yellow clarifying solution4.05 30.0 100.68 90 4.06 30.3 101.12 180 4.08 30.1 101.79 2 0 淡黄色澄清溶液
Pale yellow clarifying solution4.03 30.2 101.80 90 4.00 29.9 99.26 180 4.05 30.3 99.59 3 0 淡黄色澄清溶液
Pale yellow clarifying solution4.04 30.4 101.87 90 4.02 30.1 100.76 180 4.06 30.2 100.46 表 4 多西环素温敏型原位凝胶子宫灌注液在母猪子宫内的药物质量浓度
Table 4 Drug mass concentration of doxycycline thermo-sensitive in situ gel uterine perfusate in sow uterus
ρ/(μg·mL−1) 采样时间
Sampling time母猪编号1) Sow number ${\bar X }$±S 1 2 3 4 5 第1次给药12 h后 12 h after the first administration 85.76 0.53 102.21 269.32 48.71 101.31±101.73 第1次给药24 h后 24 h after the first administration — 191.27 18.76 258.31 115.77 116.82±110.48 第1次给药36 h后 36 h after the first administration 1.80 — 3.77 187.32 27.36 44.05±80.86 第2次给药前 Before the second administration 0.35 2.85 — 211.50 64.39 55.82±91.25 第2次给药8 h后 8 h after the second administration 477.33 8.07 167.14 163.36 104.64 184.11±176.07 第2次给药12 h后 12 h after the second administration 391.98 11.25 1870.95 86.11 4.28 472.91±797.41 第2次给药24 h后 24 h after the second administration 109.63 4.64 51.34 240.79 401.62 161.60±160.76 第2次给药36 h后 36 h after the second administration 161.18 3.91 33.14 120.06 159.39 95.54±72.93 第2次给药48 h后 48 h after the second administration 46.23 264.80 18.21 — 170.11 99.87±113.64 第2次给药72 h后 72 h after the second administration 229.61 185.04 26.94 — 202.99 128.92±107.00 第2次给药96 h后 96 h after the second administration 130.93 95.37 10.71 — 105.70 68.54±59.24 1)“—”表示未检出
1)“—” means no doxycycline detected -
[1] 吴玉辰, 郭爽, 唐光武. 规模化猪场母猪子宫内膜炎的防治[J]. 黑龙江畜牧兽医, 2012(6): 86-87. [2] 叶健毅, 兰邹然. 母猪子宫内膜炎及其防控技术[J]. 动物医学进展, 2008, 29(7): 104-108. doi: 10.3969/j.issn.1007-5038.2008.07.027 [3] 陈杖榴. 兽医药理学[M]. 北京: 中国农业出版社, 2004. [4] 廖治锋, 段新华. 兽用多西环素的研究进展[J]. 黑龙江畜牧兽医, 2015(9): 76-78. [5] 王军, 石冬梅, 陈益, 等. 多西环素泡腾片治疗子宫内膜炎和胎衣不下临床效果观察[J]. 中国兽医杂志, 2012, 48(8): 70-72. doi: 10.3969/j.issn.0529-6005.2012.08.029 [6] 舒予, 李小芳, 吴珊, 等. 原位凝胶给药系统在中药制剂中的应用研究进展[J]. 成都中医药大学学报, 2014, 37(3): 120-123. [7] GU F, FAN H, CONG Z, et al. Preparation, characterization, and in vivo pharmacokinetics of thermosensitive in situ nasal gel of donepezil hydrochloride[J]. Acta Pharmaceutica, 2020, 70(3): 411-422. doi: 10.2478/acph-2020-0032
[8] RAJPUT A P, BUTANI S B. Resveratrol anchored nanostructured lipid carrier loaded in situ gel via nasal route: Formulation, optimization and in vivo characterization[J]. Journal of Drug Delivery Science and Technology, 2019, 51: 214-223. doi: 10.1016/j.jddst.2019.01.040
[9] ABOU YOUSSEF N A H, KASSEM A A, FARID R M, et al. A novel nasal almotriptan loaded solid lipid nanoparticles in mucoadhesive in situ gel formulation for brain targeting: Preparation, characterization and in vivo evaluation[J]. International Journal of Pharmaceutics, 2018, 548(1): 609-624. doi: 10.1016/j.ijpharm.2018.07.014
[10] ALHARBI W S, HOSNY K M. Development and optimization of ocular in situ gels loaded with ciprofloxacin cubic liquid crystalline nanoparticles[J]. Journal of Drug Delivery Science and Technology, 2020, 57: 101710. doi: 10.1016/j.jddst.2020.101710.
[11] LI J, ZHAO H, OKEKE C I, et al. Comparison of systemic absorption between ofloxacin ophthalmic in situ gels and ofloxacin conventional ophthalmic solutions administration to rabbit eyes by HPLC–MS/MS[J]. International Journal of Pharmaceutics, 2013, 450(1/2): 104-113.
[12] DESHKAR S S, PALVE V K. Formulation and development of thermosensitive cyclodextrin-based in situ gel of voriconazole for vaginal delivery[J]. Journal of Drug Delivery Science and Technology, 2019, 49: 277-285. doi: 10.1016/j.jddst.2018.11.023
[13] 徐彦, 潘立群, 施义, 等. 温度敏感型盐酸川芎嗪腹腔用原位缓释凝胶的制备研究与评价[J]. 时珍国医国药, 2007, 18(5): 1068-1070. doi: 10.3969/j.issn.1008-0805.2007.05.025 [14] 中国兽药典委员会. 中华人民共和国兽药典: 第1部[M]. 北京: 中国农业出版社, 2015: 194-196. [15] 沈雪, 常笛, 姜同英, 等. 硫普罗宁眼用原位凝胶的研制[J]. 沈阳药科大学学报, 2009, 26(2): 89-92. [16] LU C, LIU M, FU H, et al. Novel thermosensitive in situ gel based on poloxamer for uterus delivery[J]. European Journal of Pharmaceutical Sciences, 2015, 77: 24-28. doi: 10.1016/j.ejps.2015.05.014
[17] CHOI H G, JUNG J H, RYU J M, et al. Development of in situ-gelling and mucoadhesive acetaminophen liquid suppository[J]. International Journal of Pharmaceutics, 1998, 165(1): 33-44. doi: 10.1016/S0378-5173(97)00386-4
[18] YONG C S, OH Y K, JUNG S H, et al. Preparation of ibuprofen-loaded liquid suppository using eutectic mixture system with menthol[J]. European Journal of Pharmaceutical Sciences, 2004, 23(4/5): 347-353.
[19] LIU Y, ZHU Y Y, WEI G, et al. Effect of carrageenan on poloxamer-based in situ gel for vaginal use: Improved in vitro and in vivo sustained-release properties[J]. European Journal of Pharmaceutical Sciences, 2009, 37(3/4): 306-312.
[20] YUAN Y, CUI Y, ZHANG L, et al. Thermosensitive and mucoadhesive in situ gel based on poloxamer as new carrier for rectal administration of nimesulide[J]. International Journal of Pharmaceutics, 2012, 430(1/2): 114-119.
[21] MENDONSA N S, MURTHY S N, HASHEMNEJAD S M, et al. Development of poloxamer gel formulations via hot-melt extrusion technology[J]. International Journal of Pharmaceutics, 2018, 537(1/2): 122-131.
[22] 国家药典委员会. 中华人民共和国药典: 第4部[M]. 北京: 中国医药科技出版社, 2015: 121-124. [23] 王丽娟, 朱照静. 泊洛沙姆407温度敏感型原位凝胶的研究进展[J]. 中国药学杂志, 2009, 44(4): 245-248. doi: 10.3321/j.issn:1001-2494.2009.04.002 [24] 周亚玉, 柴方红, 李春花, 等. 母猪子宫内膜炎的病原分离及药敏试验[J]. 吉林畜牧兽医, 2014, 35(5): 15-17. doi: 10.3969/j.issn.1672-2078.2014.05.003 [25] 叶健毅, 赵学峰, 兰邹然, 等. 规模化猪场母猪子宫内膜炎致病菌的分离鉴定[J]. 畜牧兽医杂志, 2011, 30(1): 5-7. doi: 10.3969/j.issn.1004-6704.2011.01.002 [26] 王春阳. 盐酸多西环素口服液的制备及安全性试验[D]. 杨凌: 西北农林科技大学, 2016. [27] 农业部兽药评审中心. 兽药研究技术指导原则汇编[G]. 北京: 化学工业出版社, 2012: 47-51.




 下载:
下载: