Effect of dietary supplementation of fish oil on intestinal barrier function in mouse fed with high-fat diet
-
摘要:目的
研究日粮添加鱼油对高脂日粮饲喂小鼠肠道屏障功能的影响。
方法选用36只4周龄C57BL/6J雌性小鼠,随机分为对照组、高脂组、高脂+鱼油组,每组12只小鼠,分别饲喂基础日粮、高脂日粮、高脂日粮添加质量分数为5%的鱼油(等能替换高脂日粮中脂肪)。试验持续21周,每周测定小鼠采食量和体质量,期间测定小鼠肠道通透性以及粪便粗脂肪含量和能值;试验结束后,检测小鼠血清内毒素水平,检测小鼠肠道形态、杯状细胞的数量、肠道紧密连接蛋白和炎症因子的表达。
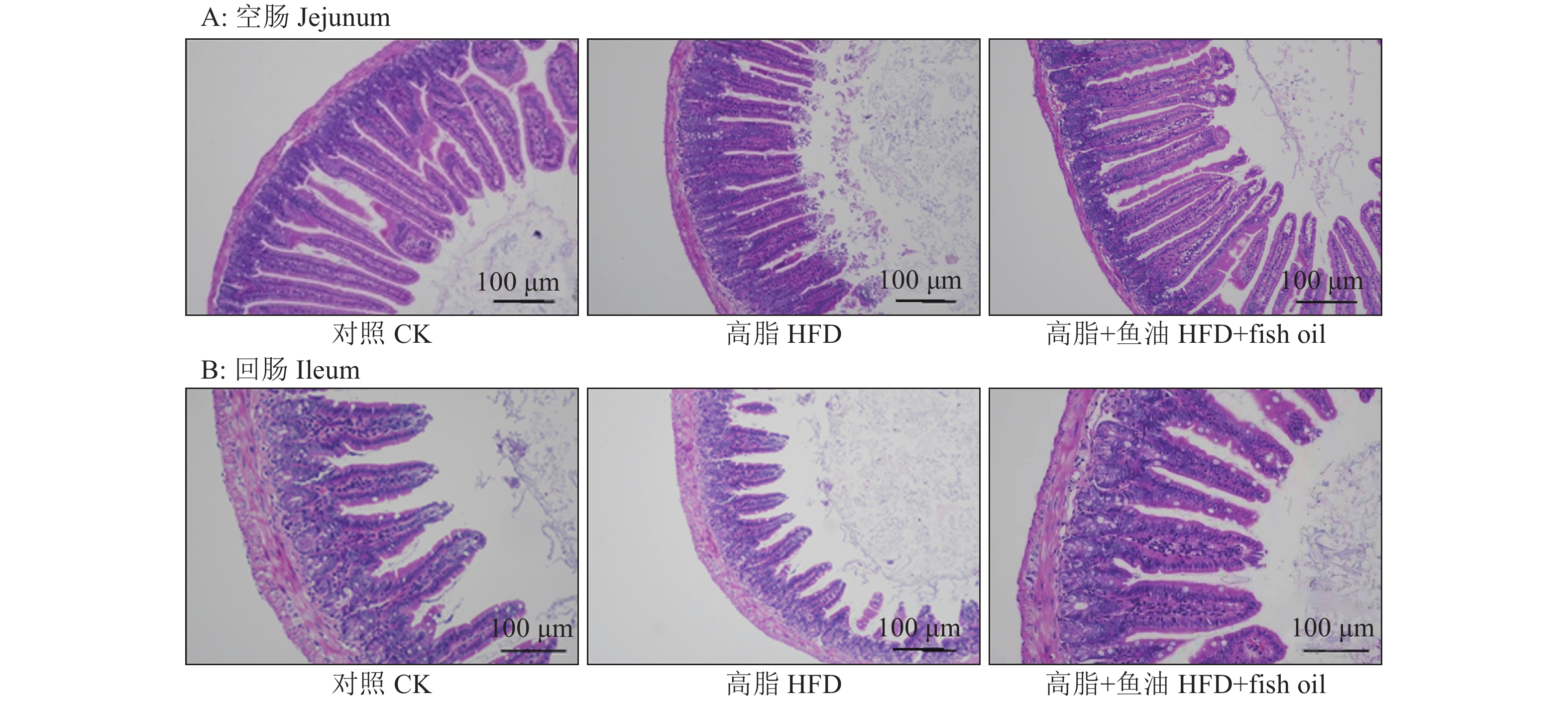
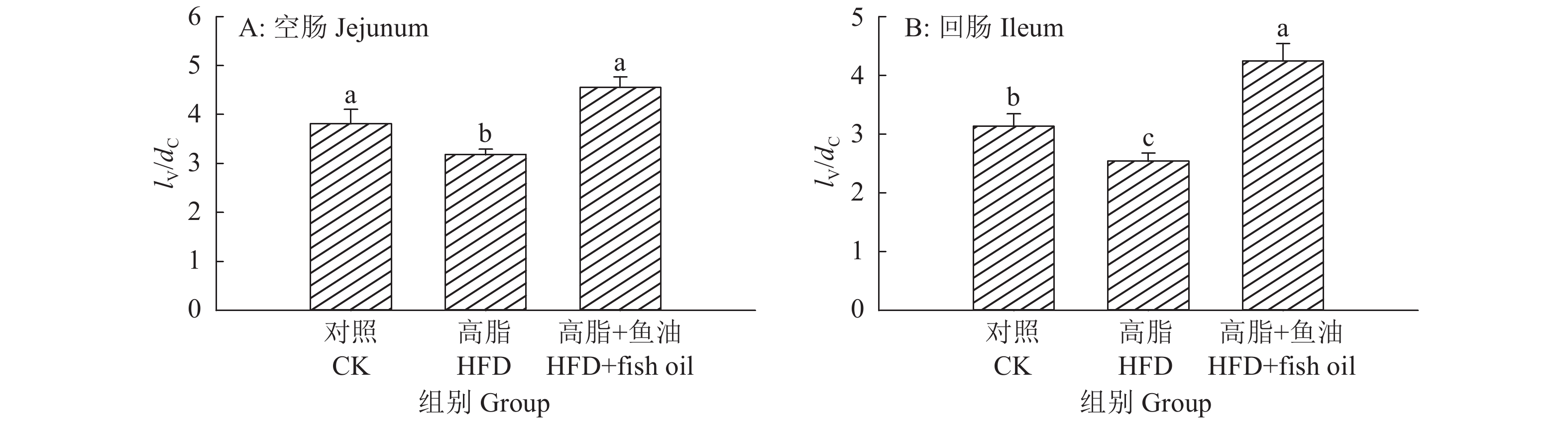
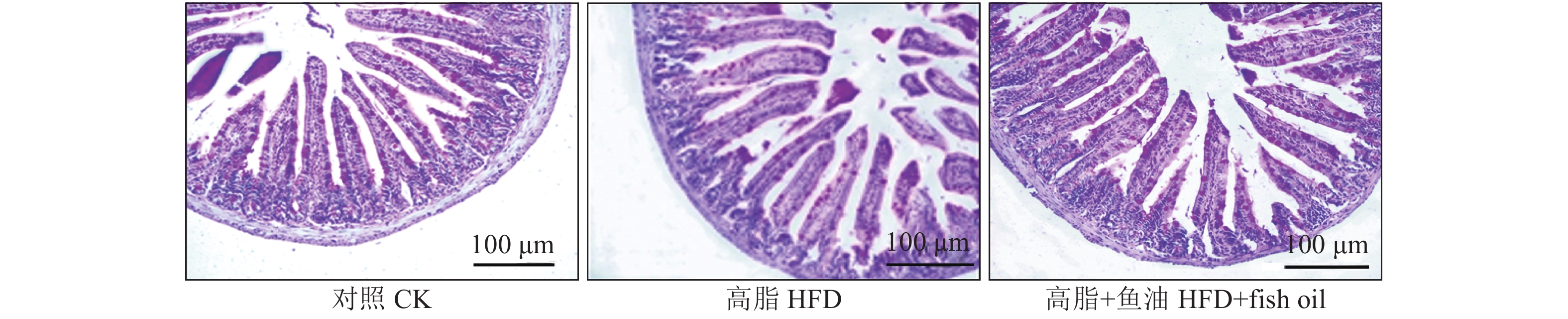
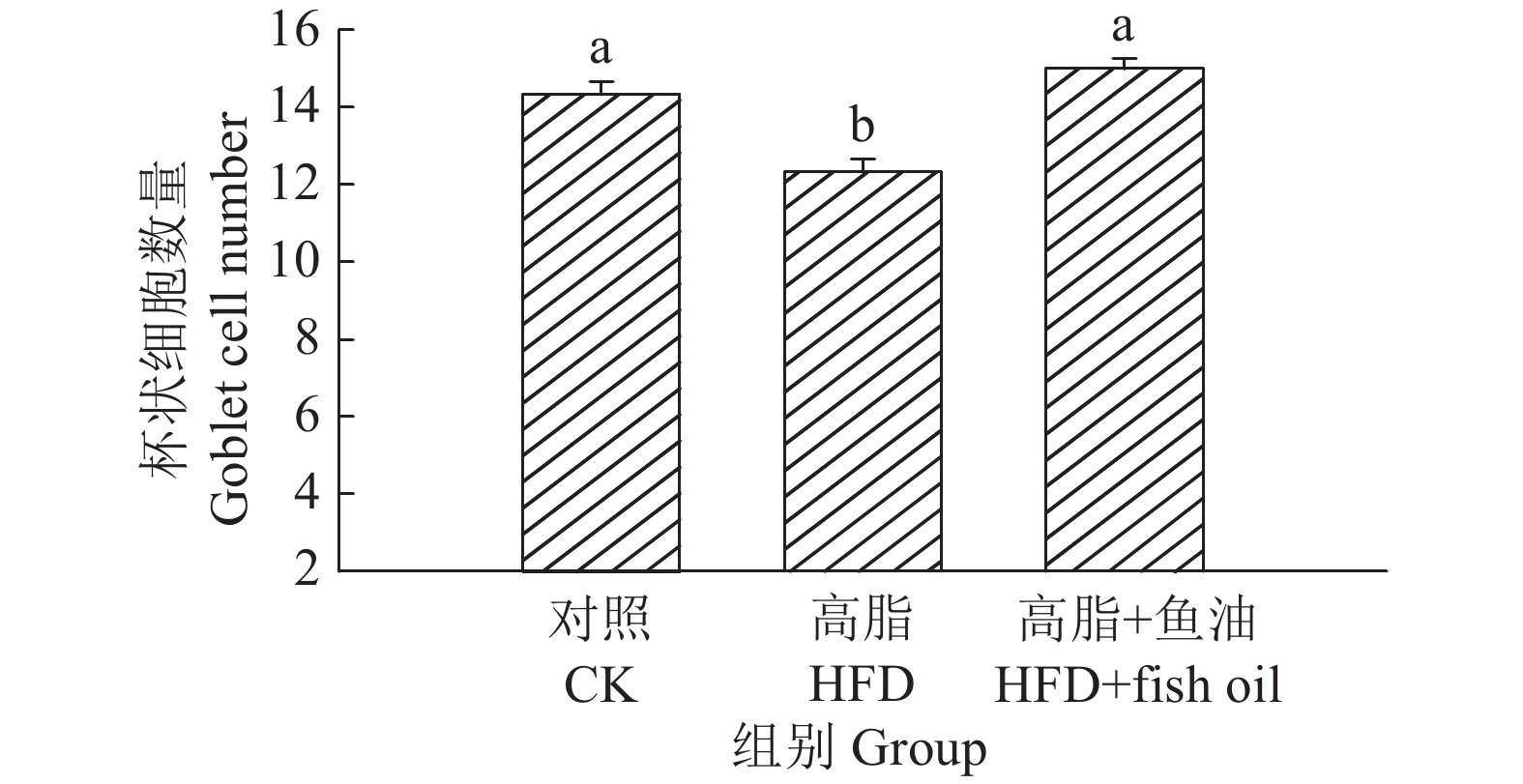
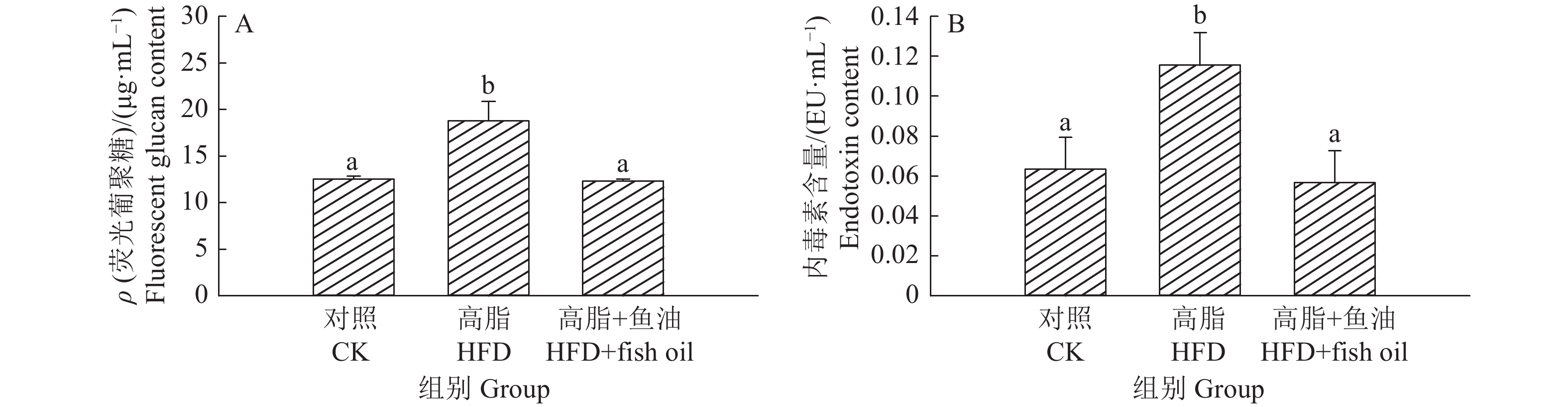
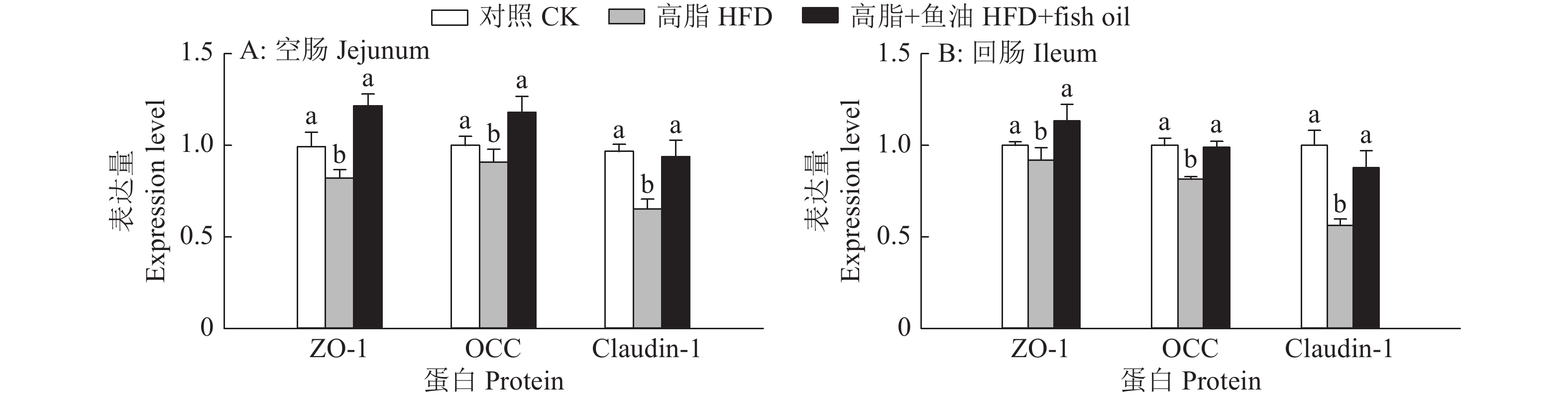
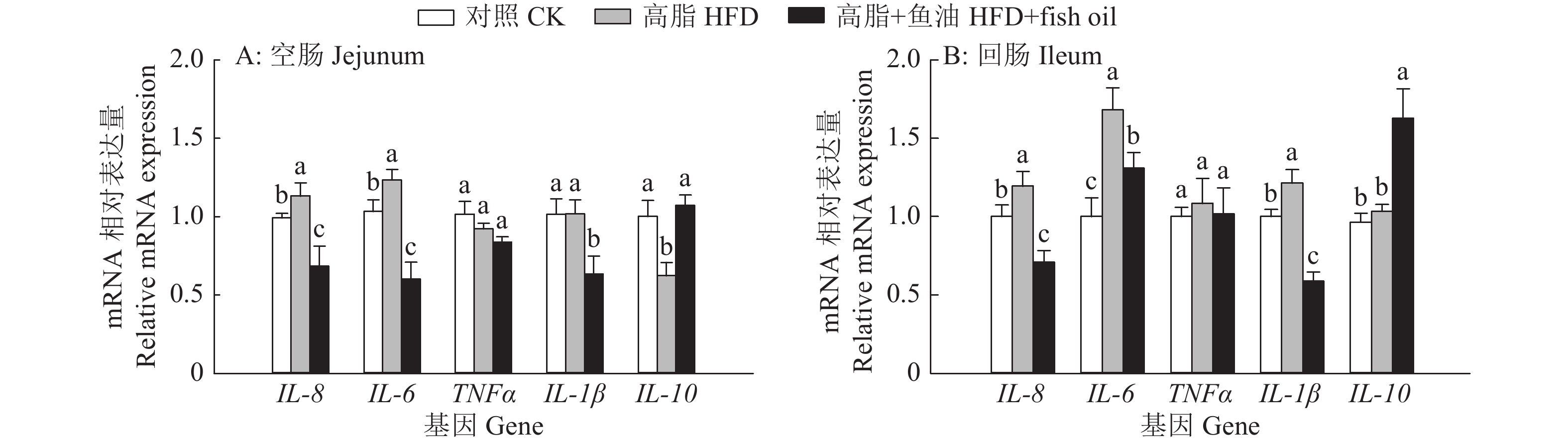
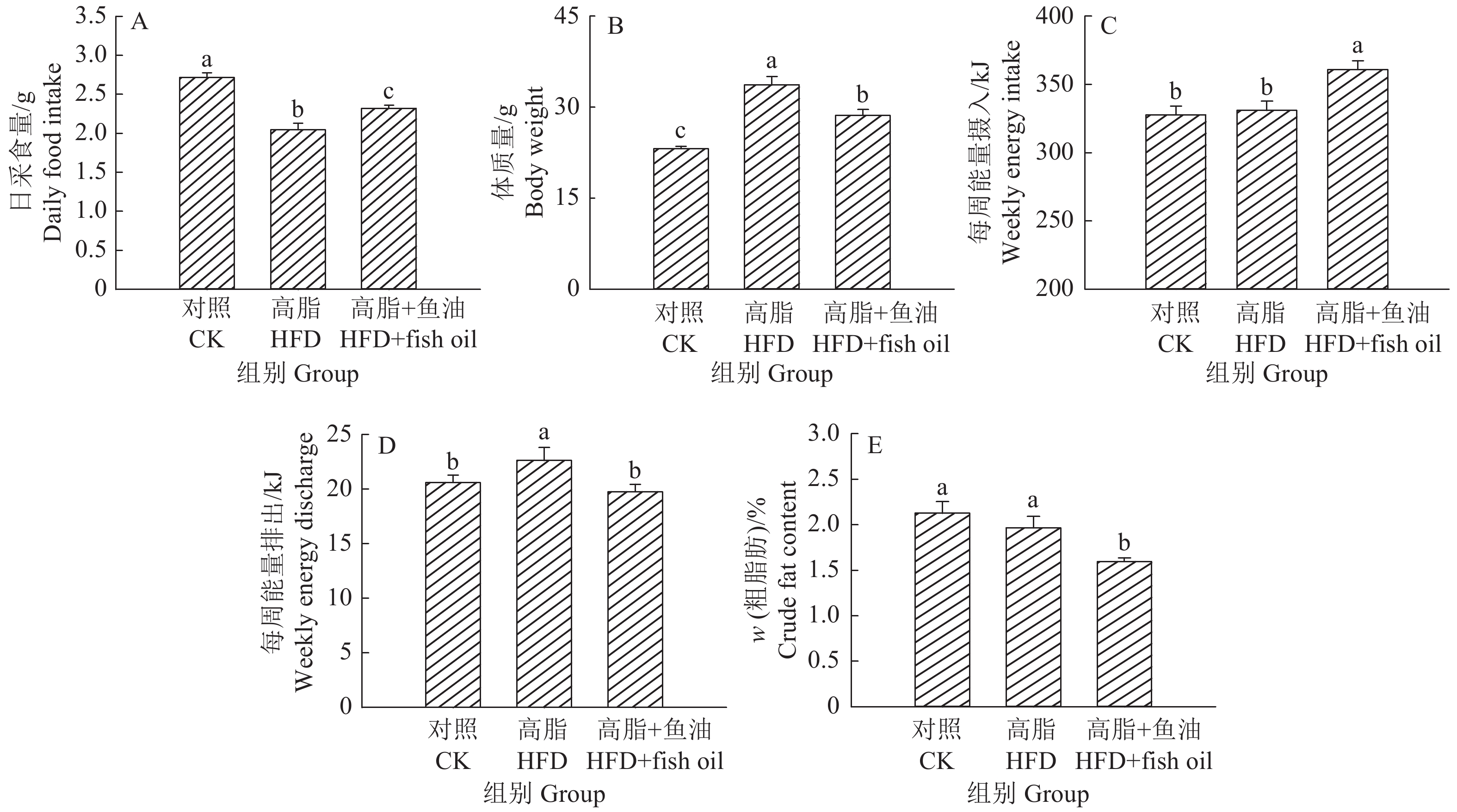
结果与高脂组相比,日粮添加鱼油显著提高了小鼠采食量和能量摄入、降低了粪便中粗脂肪的含量和能量排出,显著降低了小鼠体质量(P<0.05)。在肠道形态方面,与高脂组相比,添加鱼油使小鼠空肠和回肠的绒毛高度与隐窝深度比值(lV/dC)分别提高了43.1%和67.5%,使回肠绒毛杯状细胞的数量增多了16.7%(P<0.05)。与高脂组相比,添加鱼油使血清荧光葡聚糖和内毒素水平分别降低了34.3%和50.4%(P<0.05),并逆转了高脂日粮造成的肠道紧密连接蛋白表达的降低。在炎症因子表达方面,与高脂组相比,添加鱼油显著降低了空肠和回肠内促炎因子IL-8、IL-6和IL-1β的表达,同时显著增加了抗炎因子IL-10的表达(P<0.05)。
结论日粮添加鱼油可降低由高脂日粮导致的小鼠肠道屏障功能损伤,这可能与鱼油降低肠道炎症有关。
Abstract:ObjectiveTo investigate the effect of dietary supplementation of fish oil on the barrier function of mouse fed with high-fat diet (HFD).
Method36 C57BL/6J female mice (4-week-old) were randomly divided into control group (CK), HFD group, and HFD +fish oil group, with 12 mice in each group. Mice in three groups were fed with basal diet, HFD and HFD supplemented with 5% fish oil (iso caloric with HFD), respectively. The experiment lasted for 21 weeks, and the food intake and body weight of mice were measured weekly. During the experiment, intestinal permeability of each mouse was measured, and the fat content and energy in feces were determined. At the end of the experiment, endotoxin levels in serum were detected, and intestinal morphology, the number of intestinal goblet cells, and the expressions of intestinal tight junction proteins and inflammatory factors were examined.
ResultCompared with HFD group, addition of fish oil in diet significantly increased food intake and energy intake, decreased fecal crude fat content and energy excretion, while significantly reduced body weight of mice (P<0.05). In terms of intestinal morphology, compared with HFD group, addition of fish oil significantly increased the ratio of villus height to crypt depth (lV/dC) by 43.1% in mouse jejunum and 67.5% in ileum and the number of goblet cells in ileum villous by 16.7% (P<0.05). Compared with HFD group, addition of fish oil reduced the fluorescent glucan and endotoxin levels in serum by 34.3% and 50.4% respectively (P<0.05). Addition of fish oil also reversed the decrease in expressions of intestinal tight junction proteins caused by HFD. In terms of expression of inflammation factors, compared with HFD group, addition of fish oil significantly decreased the expressions of proinflammatory factors includingIL-8, IL-6 and IL-1β, and significantly elevated the expression of anti-inflammatory factor IL-10 in mouse jejunum and ileum (P<0.05).
ConclusionDietary supplementation of fish oil can reduce the HFD-induced impairment of intestinal barrier function, which might be associated with the fish oil reducing intestinal inflammation.
-
Keywords:
- mouse /
- fish oil /
- high-fat diet /
- intestinal barrier /
- tight junction protein /
- inflammation
-
-
图 3 日粮添加鱼油对小鼠空肠和回肠的绒毛高度与隐窝深比值(lV/dC)的影响
各图中,柱子上方的不同小写字母表示差异显著(P<0.05,Duncan’s法)
Figure 3. Effect of dietary supplementation of fish oil on the ratio of villus length to crypt depth (lV/dC) in mouse jejunum and ileum
In each graph, different lowercase letters on bars indicate significant differences(P<0.05, Duncan’s method)
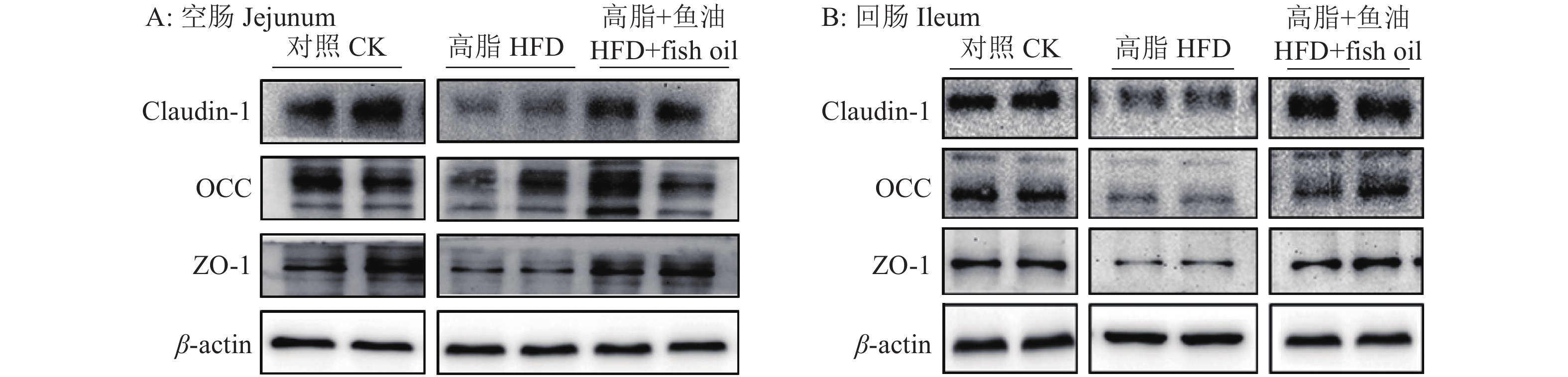
图 8 日粮添加鱼油对小鼠空肠和回肠紧密连接蛋白表达的影响
各图中,相同蛋白柱子上方的不同小写字母表示组间差异显著(P<0.05,Duncan’s法)
Figure 8. Effect of dietary supplementation of fish oil on the expression of tight junction protein in mouse jejunum and ileum
In each graph, different lowercase letters on bars of the same protein indicate significant differences among groups(P<0.05, Duncan’s method)
图 9 日粮添加鱼油对小鼠空肠和回肠炎症因子表达的影响
各图中,相同基因柱子上方的不同小写字母表示组间差异显著(P<0.05,Duncan’s法)
Figure 9. Effect of dietary supplementation of fish oil on the expression of inflammatory factor in mouse jejunum and ileum
In each graph, different lowercase letters on bars of the same gene indicate significant differences among groups(P<0.05, Duncan’s method)
表 1 小鼠日粮成分组成
Table 1 The dietary composition for mice
项目
Item组成
Component对照组
CK group高脂组
HFD group高脂+鱼油组
HFD+ fish oil group原料及质量分数/(g·kg−1)
Ingredient and content玉米淀粉 Corn starch 397.5 0 0 酪蛋白 Casein 200.0 261.7 261.7 麦芽糖糊精 Maltodextrin 132.0 163.6 163.6 蔗糖 Sucrose 100.0 89.8 89.8 豆油 Soybean oil 70.0 32.7 32.7 猪油 Lard 0 320.6 273.7 纤维素 Cellulose 50.0 65.4 65.4 混合矿物质 Mineral Mix 35.0 45.8 45.8 混合维生素 Vitamin Mix 10.0 13.1 13.1 L−胱氨酸 L−cystine 3.0 3.9 3.9 氯化胆碱 Choline chloride 2.5 3.3 3.3 鱼油 Fish Oil 0 0 50.0 合计 Total 1 000.0 999.9 1 003.0 营养成分及质量分数/%
Nutrient and content蛋白 Protein 17.9 26.2 26.2 脂肪 Fat 7.0 34.9 34.9 碳水化合物 Carbohydrate 64.4 26.3 26.3 总能/(kJ·g−1)
Total energy15.07 21.93 21.93 表 2 小鼠炎症因子引物序列
Table 2 Sequences for primers of mouse inflammation factors
基因
Gene引物序列(5′→3′)
Primer sequence产物长度/bp
Product lengthIL-1β F:TCTCTGGCCTTGGAACATAGTCT
R:CAAAGCCATCAGTGAACTCATCA145 IL-6 F:TACCACTCCCAACAGACCTG
R:CAAGTGCATCATCGTTGTTCA135 IL-8 F:CACCCTCTGTCACCTGCTCAA
R:ATGGCGCTGAGAAGACTTGGT101 IL-10 F:CGGGAAGACAATAACTGCACCC
R:CAAAGCCATCAGTGACCTAATCA130 TNF-α F:AGGCACTCCCCCAAAAGATG
R:CAAAGCCATCAGTGACCTAATCA145 β-actin F:TCTCTGTCCTTGGAACATAGTCT
R:CAAAGCCATCAGTGACCTAATCA142 -
[1] 彭凯, 吴薇, 龙蕾, 等. 断奶仔猪的腹泻研究进展[J]. 饲料研究, 2015(13): 10-15. [2] JI Y, SAKATA Y, TSO P. Nutrient-induced inflammation in the intestine[J]. Curr Opin Clin Nutr, 2011, 14(4): 315-321. doi: 10.1097/MCO.0b013e3283476e74
[3] DONG L, ZHANG Y, YANG L, et al. Effects of a high-fat diet on adipose tissue CD8+ T cells in young vs adult mice[J]. Inflammation, 2017, 40(6): 1944-1958. doi: 10.1007/s10753-017-0635-0
[4] CREMONINI E, WANG Z W, BETTAIEB A, et al. (-)-Epicatechin protects the intestinal barrier from high fat diet-induced permeabilization: Implications for steatosis and insulin resistance[J]. Redox Biol, 2018, 14: 588-599. doi: 10.1016/j.redox.2017.11.002
[5] HAMILTON M K, BOUDRY G, LEMAY D G, et al. Changes in intestinal barrier function and gut microbiota in high-fat diet-fed rats are dynamic and region dependent[J]. Am J Physiol-Gastr L, 2015, 308(10): G840-G851.
[6] DING S L, CHI M M, SCULL B P, et al. High-fat diet: Bacteria interactions promote intestinal inflammation which precedes and correlates with obesity and insulin resistance in mouse[J]. PLoS One, 2010, 5(8): e12191. doi: 10.1371/journal.pone.0012191
[7] PETERSON L W, ARTIS D. Intestinal epithelial cells: Regulators of barrier function and immune homeostasis[J]. Nat Rev Immunol, 2014, 14(3): 141-153. doi: 10.1038/nri3608
[8] BENÍTEZ-PÁEZ A, GÓMEZ DEL PULGAR E M, KJØLBÆK L, et al. Impact of dietary fiber and fat on gut microbiota re-modeling and metabolic health[J]. Trends Food Sci Technol, 2016, 57: 201-212. doi: 10.1016/j.jpgs.2016.11.001
[9] MUHLROTH A, LI K S, RØKKE G, et al. Pathways of lipid metabolism in marine algae, co-expression network, bottlenecks and candidate genes for enhanced production of EPA and DHA in species of chromista[J]. Mar Drugs, 2013, 11(11): 4662-4697. doi: 10.3390/md11114662
[10] 左正三, 郭东升, 纪晓俊, 等. 肠道中多不饱和脂肪酸及其衍生物研究进展[J]. 中国生物工程杂志, 2018, 38(11): 66-75. [11] 李启艳, 谢强胜, 刁飞燕, 等. 鱼油的化学成分及其药理活性研究进展[J]. 药物分析杂志, 2016, 36(7): 1157-1161. [12] MORI T A, BEILIN L J. Omega-3 fatty acids and inflammation[J]. Curr Atheroscler Rep, 2004, 6(6): 461-467. doi: 10.1007/s11883-004-0087-5
[13] TONTONOZ P, SPIEGELMAN B M. Fat and beyond: The diverse biology of PPARγ[J]. Annu Rev Biochem, 2008, 77(1): 289-312. doi: 10.1146/annurev.biochem.77.061307.091829
[14] 龙烁, 王浩, 武书庚, 等. 二十二碳六烯酸的生理学功能及其在家禽生产中的应用[J]. 动物营养学报, 2017, 29(4): 1101-1109. doi: 10.3969/j.issn.1006-267x.2017.04.003 [15] 唐军旺, 方倩倩, 邵荣益, 等. 不同油脂对填饲期朗德鹅生产性能、血清指标及肝脏脂肪酸组成的影响[J]. 动物营养学报, 2018, 30(7): 2550-2560. doi: 10.3969/j.issn.1006-267x.2018.07.015 [16] 银忠. 地衣芽孢杆菌对菌群失调大鼠肠道微生态、黏膜结构和免疫细胞的影响[D]. 武汉: 华中农业大学, 2009. [17] HASAN A U, OHMORI K, KONISHI K, et al. Eicosapentaenoic acid upregulates VEGF-A through both GPR120 and PPARγ mediated pathways in 3T3-L1 adipocytes[J]. Mol Cell Endocrinol, 2015, 406: 10-18. doi: 10.1016/j.mce.2015.02.012
[18] 贺显晶, 孙东波, 武瑞. 鱼油对断奶SD大鼠生产性能、脏器指数及肠道形态的影响[J]. 黑龙江畜牧兽医, 2011(9): 144-146. [19] 张智, 单庆文, 王琳琳, 等. 培菲康对高脂饮食诱导脂肪肝病大鼠肠黏膜屏障的保护作用[J]. 世界华人消化杂志, 2013, 21(2): 130-137. [20] MOREIRA A P B, TEXEIRA T F S, FERREIRA A B, et al. Influence of a high-fat diet on gut microbiota, intestinal permeability and metabolic endotoxaemia[J]. Br J Nutr, 2012, 108(5): 801-809. doi: 10.1017/S0007114512001213
[21] TAKAHASHI Y, ISUZUGAWA K, MURASE Y, et al. Up-regulation of NOD1 and NOD2 through TLR4 and TNF-α in LPS-treated murine macrophages[J]. J Vet Med Sci, 2006, 68(5): 471-478. doi: 10.1292/jvms.68.471
[22] GREGOR M F, HOTAMISLIGIL G S. Inflammatory mechanisms in obesity[J]. Annu Rev Immunol, 2011, 29(1): 415-445. doi: 10.1146/annurev-immunol-031210-101322
[23] GEURTS L, NEYRINCK A M, DELZENNE N M, et al. Gut microbiota controls adipose tissue expansion, gut barrier and glucose metabolism: Novel insights into molecular targets and interventions using prebiotics[J]. Benef Mirbobes, 2014, 5(1): 3-17. doi: 10.3920/BM2012.0065



 下载:
下载: