Progress in glucosinolates and its molecular mechanism of biosynthesis
-
摘要:
硫代葡萄糖苷(简称硫苷)是植物中一种重要的植物次生代谢物质,具有很多功能,尤其是其中的萝卜硫苷的降解产物——萝卜硫素具有抗癌作用,因此,受到广泛重视。近年来硫苷研究取得了重要进展。本文系统地介绍了硫苷的功能、种类、分布、运输、生物合成、降解、影响合成和积累的因素、基因工程,并展望其研究前景。
Abstract:Glucosinolates (GS) are important secondary metabolites in plants. They have many functions, especially the breakdown product of glucoraphanin, sulphoraphane, has anti-cancer properties and therefore has been paid great attention. A great progress has been made in research of glucosinolates in recent years. This paper presents a review on the function, type, distribution, transportation, biosynthesis and degradation of glucosinolate, factors affecting the biosynthesis and accumulation, genetic engineering, prospects and so on.
-
Keywords:
- glucosinolates /
- Crucifera /
- biosynthesis /
- degradation /
- molecular mechanism
-
首先,确定母猪具有高产的遗传品质,后备母猪培育与管理到位。这2点是决定猪场PSY的启动基础;其次,合理的胎龄结构,即第2至第5胎(或至6、7胎)的母猪数量比例是另一个关键因素。简而言之,具有高产遗传品质的后备母猪,及1、2胎产仔好是必要条件[1];顺利过渡到3~5胎为主结构的高产母猪群[2-3]是充分条件;后者成为主因时,合理的母猪群体更新率成为非常关键的因素。
选择合适的高产母猪,如丹系、法系和加系母系猪,在管理上严格要求,其中,基于最新高产母猪的正常生产指标应是第1、2胎总产仔数达到30头[4];在后备母猪发育、培育的首配标准上[5],笔者认为,关键点排序如下:
1) 体重:135~170 kg首配,这一点非常重要。一生总产仔数是在135~160 kg首配的最多,经济上也最划算。体重太大,会影响到维持需要及淘汰率、肢蹄问题。体重太小、产仔太多时易造成钙磷流失过度而淘汰。
2) 配种日龄为220~270 d,有研究报道不能超过290 d[6],众多资料表明需让母猪在1岁左右分娩,使用年限才较长;6.5~7.0月龄后,发情期刺激6周内应有90%的后备母猪呈现发情。
3) 背膘为12~18 mm,具体视品系特点而定,体况评分为3.0~3.5。后备母猪背膘控制因品系不同,高产母猪在妊娠期膘情控制应更为严格。
4) 全期日增重(初生至140 kg)为590~850 g。对当代高产母猪而言,生长速度过快过慢都会影响到使用寿命、后备利用率和产仔表现。
5) 维持合适选留率、利用率的流程管控。不是所有母猪都适合留种,各阶段后备猪选育,发情和一生繁殖循环的持续性表现好、使用寿命期长、总产仔数量很重要。在29~31周龄时,母猪选留率能大于60%。
1. 年更新率
年更新率=母猪淘汰率+母猪死亡率,其中后者包括无害化处理和自然或疾病死亡。据报道,在欧洲母猪平均年更新率为43%~52%,平均淘汰胎次为4.3~4.6;法国高产的母猪年更新率是44%[7-9];在美国,母猪的年更新率为54%,包括主动和被动淘汰以及死亡率;平均淘汰胎次为3.3~3.8[10-11]。
美国爱荷华州立大学Ken Stalder教授认为PigCHAMP和Pigtales生产管理软件系统中的更新率分别为60%和53%,淘汰率分别为42%和47%(PigCHAMP全美软件,Pigtales是PIC公司软件);并指出更新率方面,在澳大利亚是63.8%(1992—2002年),巴西2002年为55%(43%淘汰率和5.5%死亡率),加拿大2002年为58%,日本2000年为47.6%。2002 PigCHAMP系统中最好的10%猪场指标为:更新率32.7%、淘汰率22%、死亡率2.8%、淘汰平均胎次在5.5胎。因而,除了高产后备母猪的准备外,合理的胎龄结构、平均淘汰胎次是实现猪场高产的其他关键影响指标。
综合众多研究,建议更新率39%~40%为管理目标的基准,其中淘汰率35%~36%,死亡率3%~5%[12];从理论或经济上来说,胎次不超过8,提供断奶仔猪总数88头是一个管理的基准目标。
2. 淘汰率及原因剖析
能繁母猪是全猪场最有价值的动物即生物资产,据估计40%~50%母猪在第3或4胎之前被淘汰。母猪在分娩1~2胎后被动淘汰时,母猪最大生产能力未得以发挥,替换成本还未得到收回[13-14]。母猪淘汰原因中,约有70%是由于繁殖失败和肢蹄疾病,即被动淘汰是主因[9]。一头母猪在第3胎时才足够抵回/收回成本,当然实际情况因猪场、投资和国家而异[13-15]。高淘汰率增加非生产天数,增加后备母猪数量和引种比例,导致平均窝产仔数、PSY下降[16]。主动淘汰是指数据管理和现场人员决定从能繁母猪群中主动移除母猪的操作流程。原因为达到最大胎次即年老、产仔表现不佳。而被动淘汰是由于繁殖障碍、哺乳问题、肢蹄疾病及死亡。年轻母猪常为被动淘汰之列[17]。美国爱荷华州立大学Ken Stalder教授早期讲课资料中[18],分析过淘汰原因占比,其中繁殖障碍30%~35%;胎龄老化15%~20%;繁殖表现差15%~20%,肢蹄问题10%~15%,死亡5%~10%,产后瘫痪或产后继发问题3%~5%,其他占5%。从结果可看出,主动淘汰即2、3项之和也可占30%~40%,可以成为管理的目标。
在河南农业大学李新建博士等[19]翻译的《妊娠和哺乳母猪》基础上,笔者进行了主动淘汰及被动淘汰的分类。主动淘汰主要为胎龄过老、产仔差等;被动淘汰为繁殖障碍、母猪的肢蹄问题以及母猪死亡等。
因胎龄过老而主动更新的淘汰占比约为9%~31%。产仔差包括窝产仔数少,出生重过轻,断奶不行,乳房和母猪质量不行,母猪因产仔差而淘汰的平均占比为20%~30%,文献记录范围为11%~56%。发情、受胎和分娩即空怀、难产等繁殖障碍是常见的被动淘汰原因,占淘汰数量比约为20%~43%;其中,有34%~43%是胎龄较低的年轻繁殖母猪,与其他动物相比,非生产日龄延长更多。据报道,在母猪群体中有6%~40%(平均10%)的移除淘汰是由于肢蹄问题。据统计,跛行后备母猪产仔数更少,平均利用年限短。研究发现,7%的跛行后备母猪未完成第1胎分娩;13%的跛行后备母猪在第2胎前淘汰;只有约50%的跛行后备母猪生产达到了第4胎。因跛行、肢蹄问题的母猪平均只能达2.93窝的生产寿命或留场时间。肢蹄问题普遍出现于第1胎母猪[19]。肢蹄评估体系有许多研究报告,且显示遗传力为中等范围:0.01~0.47,而许多特征值的遗传力>0.15[20]。Lopez-Serrano等[21]报道1至2胎留群率与日增重遗传相关系数为-0.28 (大白),-0.06 (长白),1至2胎留群率与背膘遗传相关系数为0.22 (大白),0.24 (长白),结果说明肢蹄评分越高,1~2胎过渡留群更好,肢蹄评分和1至2胎留群率遗传相关系数为0.08(大白),0.19(长白)。此外,已有研究证实肢蹄结构完整即评分好的母猪一生产仔的总量明显更多[22]。因母猪死亡而淘汰的比例为7%~15%。但美国农业部报告认为繁殖母猪群死亡率不应超过3%。根据资料,美国农场母猪死亡率随着群体增大而增加,在250头的1个试验场死亡率为2.5%,在超过500头母猪的农场死亡率增至3.7%,母猪死亡时平均胎次是3.4~4.2[23]。
3. 降低年更新淘汰率
降低年更新淘汰率,在遗传学方面表现为提高母猪利用年限。母猪利用年限是一个复杂的特征, 以非遗传因素影响为主,即不能通过核心群对母猪利用年限进行直接选育,基本无遗传选择反应。研究[7, 11, 14, 18]认为繁殖障碍或不能发情、不能配种成功是母猪淘汰的首要原因,肢蹄问题是年轻母猪淘汰的第二主因。针对这些原因,可以制定相应的措施降低淘汰率。
繁殖障碍一般可通过饲喂管理、体况标准、繁殖知识及现场管理来改变。提高后备母猪的选择标准是施行的主要措施之一,即通过产仔母猪自身的严选来预计性地提高使用年限,相应地降低早期淘汰率。
肢蹄结构的遗传力为中等偏低[24],选择具有最佳形态特征的肢蹄结构的后备母猪是防止淘汰的重点。趾、蹄病变会造成母猪的直接淘汰,母猪的非正常生长会引起淘汰风险[25],而且母猪窝产仔数量比正常母猪更少。选择肢蹄完整良好的结构选育是母猪利用年限指标的间接选育法。
此外,应关注营养。注意后备母猪培育营养、经产母猪体况饲喂及添加剂的使用;及时根据研究进展总结后对母猪进行科学管理;减轻疾病如蓝耳病对使用寿命的不利影响等。
-
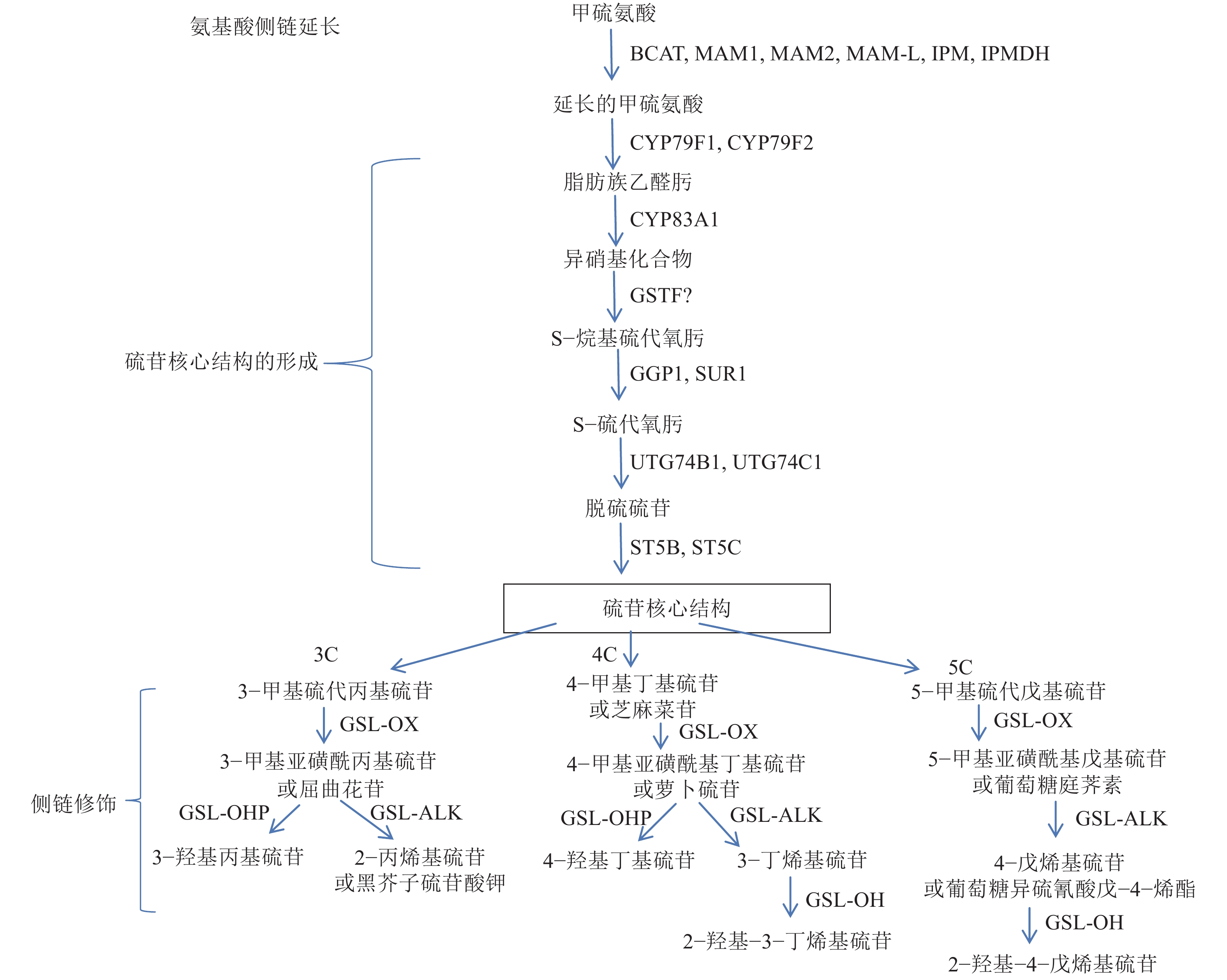
图 1 脂肪族硫苷合成路径(根据Augustine等[31]修改)
方向线旁边的为催化合成的酶,方向线所指的为产物;3C、4C和5C分别指含3、4和5个碳原子
Figure 1. General scheme for aliphatic glucosinolate biosynthesis (Modified based on Augustine et al[31])
The compound beside the direction line is catalytic enzyme, and the compound that the direction line points to is the reaction product; 3C, 4C and 5C represent compounds with 3, 4 and 5 carbon atoms respectively
-
[1] 彭佩, 卿志星, 田艳, 等. 十字花科植物中硫代葡萄糖苷激发因子研究进展[J]. 食品安全质量检测学报, 2019, 10(4): 886-891. [2] 魏大勇, 崔艺馨, 熊清, 等. 用全基因组关联作图和共表达网络分析鉴定油菜种子硫苷含量的候选基因[J]. 作物学报, 2018, 44(5): 5-17. [3] 毛舒香, 王军伟, 徐浩然, 等. 十字花科蔬菜萝卜硫素合成代谢相关基因及外源调控[J]. 中国细胞生物学学报, 2018, 40(8): 1415-1423. doi: 10.11844/cjcb.2018.08.0376 [4] SCHONHOF I, KRUMBEIN A, BRÜCKNER B. Genotypic effects on glucosinolates and sensory properties of broccoli and cauliflower[J]. Mol Nutr Food Res, 2010, 48(1): 25-33.
[5] PADILLA G, CARTEA M E, VELASCO P, et al. Variation of glucosinolates in vegetable crops of Brassica rapa[J]. Phytochemistry, 2007, 68(4): 536-545. doi: 10.1016/j.phytochem.2006.11.017
[6] ABUYUSUF M, ROBIN A, LEE J, et al. Glucosinolate profiling and expression analysis of glucosinolate biosynthesis genes differentiate white mold resistant and susceptible cabbage lines[J]. Int J Mol Sci, 2018, 19(12): 4037. doi: 10.3390/ijms19124037
[7] AIRES A, CARVALHO R, BARBOSA M D C, et al. Suppressing potato cyst nematode, Globodera rostochiensis, with extracts of Brassicacea plants[J]. Am J Potato Res, 2009, 86(4): 327-333. doi: 10.1007/s12230-009-9086-y
[8] LUCARINI E, MICHELI L, TRALLORI E, et al. Effect of glucoraphanin and sulforaphane against chemotherapy‐induced neuropathic pain: Kv7 potassium channels modulation by H2S release in vivo[J]. Phytother Res, 2018, 32(11): 2226-2234. doi: 10.1002/ptr.v32.11
[9] 罗丽娜. 硫代葡萄糖苷水解产物抗肿瘤作用的研究[D]. 武汉: 华中科技大学, 2007. [10] LIU B, MAO Q Q, CAO M, et al. Cruciferous vegetables intake and risk of prostate cancer: A meta-analysis[J]. INT J Urol, 2012, 19(2): 134-141. doi: 10.1111/iju.2012.19.issue-2
[11] WU Q J, YANG Y, VOGTMANN E, et al. Cruciferous vegetables intake and the risk of colorectal cancer: A meta-analysis of observational studies[J]. Ann Oncol, 2013, 24(4): 1079-1087. doi: 10.1093/annonc/mds601
[12] LIU X, LÜ K. Cruciferous vegetables intake is inversely associated with risk of breast cancer: A meta-analysis[J]. Breast, 2013, 22(3): 309-313. doi: 10.1016/j.breast.2012.07.013
[13] CITI V, PIRAGINE E, PAGNOTTA E, et al. Anticancer properties of erucin, an H2S-releasing isothiocyanate, on human pancreatic adenocarcinoma cells (AsPC-1)[J]. Phytother Res, 2019, 33(3): 845-855. doi: 10.1002/ptr.v33.3
[14] ABBAOUI B, LUCAS C, RIEDL K M, et al. Cruciferous vegetables, isothiocyanates and bladder cancer prevention[J]. Mol Nutr Food Res, 2018,62(18). doi: 10.1002/mnfr.20180079.
[15] MAWSON R, HEANEY R K, ZDUNCZYK Z, et al. Rapeseed meal-glucosinolates and their antinutritional effects: Part 6: Taint in end-products[J]. Die Nahrung, 2010, 39(1): 21-31.
[16] FAHEY J W, ZALCMANN A T, TALALAY P. The chemical diversity and distribution of glucosinolates and isothiocyanates among plants[J]. Phytochemistry, 2001, 56(1): 5-51. doi: 10.1016/S0031-9422(00)00316-2
[17] CLARKE D B. Glucosinolates, structures and analysis in food[J]. Anal Methods-UK, 2010, 2(4): 310-325. doi: 10.1039/b9ay00280d
[18] AGERBIRK N, OLSEN C E. Glucosinolate structures in evolution[J]. Phytochemistry, 2012, 77(1): 16-45.
[19] HWANG I M, PARK B, DANG Y M, et al. Simultaneous direct determination of 15 glucosinolates in eight Brassica species by UHPLC-Q-Orbitrap-MS[J]. Food Chem, 2019, 282: 127-133. doi: 10.1016/j.foodchem.2018.12.036
[20] LEI J, CHEN G, CHEN C, et al. Germplasm diversity of Chinese kale in China[J]. Hort Plant J, 2017, 3(3): 14-17.
[21] 仙园园, 张华, 吴增宝, 等. 维药刺山柑果实硫代葡萄糖苷酶解条件的研究及产物的鉴定[J]. 中国医院药学杂志, 2014, 34(15): 1255-1259. [22] 李文钊, 杜依登, 朱华平, 等. 响应面试验优化番木瓜籽中硫代葡萄糖苷酶解工艺[J]. 食品科学, 2014, 35(18): 28-31. doi: 10.7506/spkx1002-6630-201418006 [23] 司雨, 陈国菊, 雷建军, 等. 不同基因型芥蓝硫代葡萄糖苷组分与含量分析[J]. 中国蔬菜, 2009, 1(6): 7-13. [24] QIAN H, SUN B, MIAO H, et al. Variation of glucosinolates and quinone reductase activity among different varieties of Chinese kale and improvement of glucoraphanin by metabolic engineering[J]. Food Chem, 2015, 168(168): 321-326.
[25] OERLEMANS K, BARRETT D M, SUADES C B, et al. Thermal degradation of glucosinolates in red cabbage[J]. Food Chem, 2006, 95(1): 19-29. doi: 10.1016/j.foodchem.2004.12.013
[26] 江定, 陈国菊, 雷建军, 等. 硫代葡萄糖苷运输的生理生化及分子机理研究进展[J]. 植物生理学报, 2017(1): 29-37. [27] ANDERSEN T G, NOUR-ELDIN H H, FULLER V L, et al. Integration of biosynthesis and long-distance transport establish organ-specific glucosinolate profiles in vegetative Arabidopsis[J]. Plant Cell, 2013, 25(8): 3133-3145. doi: 10.1105/tpc.113.110890
[28] ANDERSEN T G, HALKIER B A. Upon bolting the GTR1 and GTR2 transporters mediate transport of glucosinolates to the inflorescence rather than roots[J]. Plant Signal Behav, 2014, 9(1): e27740.
[29] NOUR-ELDIN H H, ANDERSEN T G, BUROW M, et al. NRT/PTR transporters are essential for translocation of glucosinolate defence compounds to seeds[J]. Nature, 2012, 488(7412): 531-534. doi: 10.1038/nature11285
[30] JIANG D, LEI J, CAO B, et al. Molecular cloning and characterization of three glucosinolate transporter (GTR) genes from Chinese kale[J]. Genes, 2019, 10(3): 202. doi: 10.3390/genes10030202
[31] AUGUSTINE R, BISHT N C. Biofortification of oilseed Brassica juncea with the anti-cancer compound glucoraphanin by suppressing GSL-ALK gene family[J]. Sci Rep-UK, 2015, 5: 18005.
[32] HALKIER B A, DU L. The biosynthesis of glucosinolates[J]. Trends Plant Sci, 1997, 2(11): 425-431. doi: 10.1016/S1360-1385(97)90026-1
[33] ZHANG J, WANG H, LIU Z, et al. A naturally occurring variation in the BrMAM-3 gene is associated with aliphatic glucosinolate accumulation in Brassica rapa leaves[J]. Hort Res, 2018, 5(1): 69. doi: 10.1038/s41438-018-0074-6
[34] 杜海, 冉凤, 刘静, 等. 拟南芥硫苷生物合成相关基因的组织和胁迫诱导表达谱的全基因组分析[J]. 中国农业科学, 2016, 49(15): 2879-2897. doi: 10.3864/j.issn.0578-1752.2016.15.003 [35] TAMARA G, BETTINA B, HANS-PETER M, et al. The transcription factor HIG1/MYB51 regulates indolic glucosinolate biosynthesis in Arabidopsis thaliana[J]. Plant J, 2010, 50(5): 886-901.
[36] GIGOLASHVILI T, BERGER B, FLÜGGE U I. Specific and coordinated control of indolic and aliphatic glucosinolate biosynthesis by R2R3-MYB transcription factors in Arabidopsis thaliana[J]. Phytochem Rev, 2009, 8(1): 3-13. doi: 10.1007/s11101-008-9112-6
[37] CAI C, YUAN W, MIAO H, et al. Functional characterization of BoaMYB51s as central regulators of indole glucosinolate biosynthesis in Brassica oleracea var. alboglabra Bailey[J]. Front Plant Sci, 2018.
[38] YU Q, HAO G, ZHOU J, et al. Identification and expression pattern analysis of BoMYB51 involved in indolic glucosinolate biosynthesis from broccoli (Brassica oleracea var. italica)[J]. Biochem Bioph Res Co, 2018, 501(2): 598-604. doi: 10.1016/j.bbrc.2018.05.058
[39] SONDERBY I E, HANSEN B G, BJARNHOLT N, et al. A systems biology approach identifies a R2R3 MYB gene subfamily with distinct and overlapping functions in regulation of aliphatic glucosinolates[J]. PLoS One, 2007, 2(12): e1322. doi: 10.1371/journal.pone.0001322
[40] SONDERBY I E, BUROW M, ROWE H C, et al. A complex interplay of three R2R3 MYB transcription factors determines the profile of aliphatic glucosinolates in Arabidopsis[J]. Plant Physiol, 2010, 153(1): 348-363. doi: 10.1104/pp.109.149286
[41] SERGEY M, EYAL B, HADAR L, et al. The transcript and metabolite networks affected by the two clades of Arabidopsis glucosinolate biosynthesis regulators[J]. Plant Physiol, 2008, 148(4): 2021-2049. doi: 10.1104/pp.108.124784
[42] ARAKI R, HASUMI A, NISHIZAWA O I, et al. Novel bioresources for studies of Brassica oleracea: Identification of a kale MYB transcription factor responsible for glucosinolate production[J]. Plant Biotechnol J, 2013, 11(8): 1017-1027. doi: 10.1111/pbi.12095
[43] FENWICK G R, HEANEY R K, MULLIN W J. Glucosinolates and their breakdown products in food and food plants[J]. Crit Rev Food Sci Nutr, 1983, 18(2): 123-201. doi: 10.1080/10408398209527361
[44] BRANDT S, FACHINGER S, TOHGE T, et al. Extended darkness induces internal turnover of glucosinolates in Arabidopsis thaliana leaves[J]. PLoS One, 2018, 13(8): e202153.
[45] LIANG H, WEI Y, LI R, et al. Intensifying sulforaphane formation in broccoli sprouts by using other cruciferous sprouts additions[J]. Food Sci Biotechnol, 2018, 27(4): 1-6.
[46] MACLEOD A J, PANESAR S S, GIL V. Thermal degradation of glucosinolates[J]. Phytochemistry, 1981, 20(5): 977-980. doi: 10.1016/0031-9422(81)83011-7
[47] MACLEOD A J, ROSSITER J T. Non-enzymic degradation of 2-hydroxybut-3-enylglucosinolate (progoitrin)[J]. Phytochemistry, 1986, 25(4): 855-858. doi: 10.1016/0031-9422(86)80016-4
[48] SYLVIE C, LAURENT D, GÉRARD B, et al. Isolation and structure elucidation of a new thermal breakdown product of glucobrassicin, the parent indole glucosinolate[J]. J Agr Food Chem, 2002, 50(18): 5185-5190. doi: 10.1021/jf020125i
[49] SONG L, THORNALLEY P J. Effect of storage, processing and cooking on glucosinolate content of Brassica vegetables[J]. Food Chem Toxicol, 2007, 45(2): 216-224. doi: 10.1016/j.fct.2006.07.021
[50] RUNGAPAMESTRY V, DUNCAN A J, FULLER Z, et al. Changes in glucosinolate concentrations, myrosinase activity, and production of metabolites of glucosinolates in cabbage (Brassica oleracea var. capitata) cooked for different durations[J]. J Agric Food Chem, 2006, 54(20): 7628-7634. doi: 10.1021/jf0607314
[51] TABART J, PINCEMAIL J, KEVERS C, et al. Processing effects on antioxidant, glucosinolate, and sulforaphane contents in broccoli and red cabbage[J]. Eur Food Res Technol, 2018, 244(4): 2085-2094.
[52] OKUNADE O, NIRANJAN K, GHAWI S K, et al. Supplementation of the diet by exogenous myrosinase via mustard seeds to increase the bioavailability of sulforaphane in healthy human subjects after the consumption of cooked broccoli[J]. Mol Nutr Food Res, 2018, 62(18): e1700980.
[53] GRONOWITZ S, SVENSSON L, OHLSON R. Studies of some nonenzymic reactions of progoitrin[J]. J Agr Food Chem, 2002, 26(4): 887-890.
[54] PALOP M L, SMITHS J P, BRINK B T. Degradation of sinigrin by Lactobacillus agilis strain R16[J]. Int J Food Microbiol, 1995, 26(2): 219-229. doi: 10.1016/0168-1605(95)00123-2
[55] SOENGAS P, CARTEA M E, VELASCO P, et al. Brassica glucosinolate rhythmicity in response to light-dark entrainment cycles is cultivar-dependent[J]. Plant Sci, 2018, 275: 28-35. doi: 10.1016/j.plantsci.2018.07.009
[56] CHEN G J, YU S, CAO B H, et al. Analysis of combining ability and heredity parameters of glucosinolates in Chinese kale[J]. [J]Afr J Biotechnol, 2010, 9(53): 9026-9031.
[57] KUSHAD M M, BROWN A F, KURILICH A C, et al. Variation of glucosinolates in vegetable crops of Brassica oleracea[J]. J Agr Food Chem, 1999, 47(4): 1541-1548. doi: 10.1021/jf980985s
[58] LENZI M, FIMOGNARI C, HRELIA P. Sulforaphane as a promising molecule for fighting cancer[J]. Mutat Res-Rev Mutat, 2007, 635(2): 90-104.
[59] DIAS J S. Nutritional quality and health benefits of vegetables: A review[J]. Food and Nutri Sci, 2012, 3(10): 1354.
[60] 马永华, 陈文妃, 陈凌云, 等. 小白菜不同叶期及不同叶位硫苷的质量摩尔浓度[J]. 浙江农林大学学报, 2018, 35(6): 132-136. [61] BELLOSTAS N, KACHLICKI P, SØRENSEN J C, et al. Glucosinolate profiling of seeds and sprouts of B. oleracea varieties used for food[J]. Sci Hortic-Amsterdam, 2007, 114(4): 234-242. doi: 10.1016/j.scienta.2007.06.015
[62] PEREIRA F M, ROSA E, FAHEY J W, et al. Influence of temperature and ontogeny on the levels of glucosinolates in broccoli (Brassica oleracea var. italica) sprouts and their effect on the induction of mammalian phase 2 enzymes[J]. J Agric Food Chem, 2002, 50(21): 6239-6244. doi: 10.1021/jf020309x
[63] RYCHLIK M, ADAM S T. Glucosinolate and folate content in sprouted broccoli seeds[J]. Eur Food Res Technol, 2008, 226(5): 1057-1064. doi: 10.1007/s00217-007-0631-y
[64] MARTINEZ-VILLALUENGA C, PEÑAS E, CISKA E, et al. Time dependence of bioactive compounds and antioxidant capacity during germination of different cultivars of broccoli and radish seeds[J]. Food Chem, 2010, 120(3): 710-716. doi: 10.1016/j.foodchem.2009.10.067
[65] TIAN Q, ROSSELOT R A, SCHWARTZ S J. Quantitative determination of intact glucosinolates in broccoli, broccoli sprouts, brussels sprouts, and cauliflower by high-performance liquid chromatography-electrospray ionization-tandem mass spectrometry[J]. Anal Biochem, 2005, 343(1): 93-99. doi: 10.1016/j.ab.2005.04.045
[66] HANLON P R, BARNES D M. Phytochemical composition and biological activity of 8 varieties of radish (Raphanus sativus L.) sprouts and mature taproots[J]. J Food Sci, 2011, 76(1): C185-C192. doi: 10.1111/j.1750-3841.2010.01972.x
[67] 郭丽萍, 朱英莲, 唐娟. 十字花科芽苗菜与成熟蔬菜生物活性成分的比较[J]. 营养学报, 2017(6): 588-593. doi: 10.3969/j.issn.0512-7955.2017.06.014 [68] RANGKADILOK N, NICOLAS M E, BENNETT R N, et al. Developmental changes of sinigrin and glucoraphanin in three Brassica species (Brassica nigra, Brassica juncea and Brassica oleracea var. italica)[J]. Sci Hortic-Amsterdam, 2002, 96(1/2/3/4): 11-26.
[69] SHIM J Y, KIM H Y, KIM D G, et al. Optimizing growth conditions for glucosinolate production in Chinese cabbage[J]. Hortic Environ Biote, 2018, 59(5): 649-657. doi: 10.1007/s13580-018-0084-1
[70] BOHINC T, TRDAN S. Environmental factors affecting the glucosinolate content in Brassicaceae[J]. J Food Agric Environ, 2012, 10: 357-360.
[71] KISSEN R, EBERL F, WINGE P, et al. Effect of growth temperature on glucosinolate profiles in Arabidopsis thaliana accessions[J]. Phytochemistry, 2016, 130: 106-118. doi: 10.1016/j.phytochem.2016.06.003
[72] ZHENG Y J, ZHANG Y T, LIU H C, et al. Supplemental blue light increases growth and quality of greenhouse pak choi depending on cultivar and supplemental light intensity[J]. J Integr Agr, 2018, 17(10): 2245-2256. doi: 10.1016/S2095-3119(18)62064-7
[73] GROENBAEK M, TYBIRK E, KRISTENSEN H L. Glucosinolate and carotenoid content of white- and yellow-flowering rapeseed grown for human consumption as sprouts and seedlings under light emitting diodes[J]. Eur Food Res Technol, 2018, 244(6): 1121-1131. doi: 10.1007/s00217-017-3027-7
[74] KOPSELL D A, SAMS C E, BARICKMAN T C, et al. Sprouting broccoli accumulate higher concentrations of nutritionally important metabolites under narrow-band light-emitting diode lighting[J]. J Am Soc Hortic Sci, 2014, 139(4): 469-477. doi: 10.21273/JASHS.139.4.469
[75] BOOTH E J, WALKER K C, GRIFFITHS D W. A time-course study of the effect of sulphur on glucosinolates in oilseed rape (Brassica napus) from the vegetative stage to maturity[J]. J Sci Food Agr, 2010, 56(4): 479-493.
[76] HU K L, ZHU Z J, ZANG Y X, et al. Accumulation of glucosinolates and nutrients in pakchoi (Brassica campestris L. ssp. chinensis var. communis ) two cultivar plants exposed to sulfur deficiency[J]. Hortic Environ Biote, 2011, 52(2): 121-127. doi: 10.1007/s13580-011-0097-5
[77] 田璐, 吴嘉琪, 李昕悦, 等. NaCl与CaCl2处理对西兰花芽苗硫苷和异硫氰酸盐含量的影响[J]. 南京农业大学学报, 2017, 40(2): 352-358. doi: 10.7685/jnau.201610002 [78] 颜廷帅, 陈日远, 杨伟, 等. 叶面喷施甲硫氨酸对白菜生长和硫苷含量的影响[J]. 河北农业科学, 2018, 22(4): 51-52. [79] KUSZNIEREWICZ B, BĄCZEK-KWINTA R, BARTOSZEK A, et al. The dose-dependent influence of zinc and cadmium contamination of soil on their uptake and glucosinolate content in white cabbage (Brassica oleracea var. capitata f. alba)[J]. Environ Toxicol Chem, 2012, 31(11): 2482-2489. doi: 10.1002/etc.v31.11
[80] 初婷, 彭畅, 郭丽萍. MgSO4处理对西兰花芽苗菜生理活性物质和抗氧化能力的影响[J]. 食品科学, 2018, 39(11): 53-59. doi: 10.7506/spkx1002-6630-201811009 [81] SÁNCHEZ-PUJANTE P J, SABATER-JARA A B, BELCHÍ-NAVARRO S, et al. Increased glucosinolate production in Brassica oleracea var. italica cell cultures due to coronatine activated genes involved in glucosinolate biosynthesis[J]. J Agr Food Chem, 2019, 67(1): 102-111. doi: 10.1021/acs.jafc.8b04298
[82] CHUNG I, REKHA K, RAJAKUMAR G, et al. Influence of silver nanoparticles on the enhancement and transcriptional changes of glucosinolates and phenolic compounds in genetically transformed root cultures of Brassica rapa ssp. rapa[J]. Bioproc Biosyst Eng, 2018, 41(11): 1665-1677. doi: 10.1007/s00449-018-1991-3
[83] TIAN M, YANG Y, ÁVILA F W, et al. Effects of selenium supplementation on glucosinolate biosynthesis in broccoli[J]. J Agr Food Chem, 2018, 66(30): 8036-8044. doi: 10.1021/acs.jafc.8b03396
[84] TROLOVE S N, YONG T, MORRISON S C, et al. Development of a method for producing selenium-enriched radish sprouts[J]. LWT-Food Sci Technol, 2018, 95: 187-192. doi: 10.1016/j.lwt.2018.04.048
[85] DI GIOIA F, AVATO P, SERIO F, et al. Glucosinolate profile of Eruca sativa, Diplotaxis tenuifolia and Diplotaxis erucoides grown in soil and soilless systems[J]. J Food Compos Anal, 2018, 69: 197-204. doi: 10.1016/j.jfca.2018.01.022
[86] ABUYUSUF M, ROBIN A, KIM H T, et al. Altered glucosinolate profiles and expression of glucosinolate biosynthesis genes in ringspot-resistant and susceptible cabbage lines[J]. Int J Mol Sci, 2018, 19(9): 2833.
[87] COCETTA G, MISHRA S, RAFFAELLI A, et al. Effect of heat root stress and high salinity on glucosinolates metabolism in wild rocket[J]. J Plant Physiol, 2018, 231: 261-270. doi: 10.1016/j.jplph.2018.10.003
[88] LÓPEZ-BERENGUER C, MARTÍNEZ-BALLESTA M D C, MORENO D A, et al. Growing hardier crops for better health: Salinity tolerance and the nutritional value of broccoli[J]. J Agr Food Chem, 2009, 57(2): 572-578. doi: 10.1021/jf802994p
[89] YUAN G, WANG X, GUO R, et al. Effect of salt stress on phenolic compounds, glucosinolates, myrosinase and antioxidant activity in radish sprouts[J]. Food Chem, 2010, 121(4): 1014-1019. doi: 10.1016/j.foodchem.2010.01.040
[90] PÉREZ-BALIBREA S, MORENO D A, GARCÍA-VIGUERA C. Improving the phytochemical composition of broccoli sprouts by elicitation[J]. Food Chem, 2011, 129(1): 35-44. doi: 10.1016/j.foodchem.2011.03.049
[91] VALE A P, SANTOS J, BRITO N V, et al. Evaluating the impact of sprouting conditions on the glucosinolate content of Brassica oleracea sprouts[J]. Phytochemistry, 2015, 115(1): 252-260.
[92] GUO R F, YUAN G F, WANG Q M. Sucrose enhances the accumulation of anthocyanins and glucosinolates in broccoli sprouts[J]. Food Chem, 2011, 129(3): 1080-1087. doi: 10.1016/j.foodchem.2011.05.078
[93] GUO R F, YUAN G F, WANG Q M. Effect of sucrose and mannitol on the accumulation of health-promoting compounds and the activity of metabolic enzymes in broccoli sprouts[J]. Sci Hortic-Amsterdam, 2011, 128(3): 159-165. doi: 10.1016/j.scienta.2011.01.014
[94] GUO R, HOU Q, YUAN G, et al. Effect of 2, 4-epibrassinolide on main health-promoting compounds in broccoli sprouts[J]. LWT-Food Sci Technol, 2014, 58(1): 287-292. doi: 10.1016/j.lwt.2014.02.047
[95] FARD N S, ABAD H H S, RAD A H S, et al. Effect of drought stress on qualitative characteristics of canola cultivars in winter cultivation[J]. Ind Crop Prod, 2018, 114: 87-92. doi: 10.1016/j.indcrop.2018.01.082
[96] EOM S, BAEK S, KIM J, et al. Transcriptome analysis in Chinese cabbage (Brassica rapa ssp. pekinensis) provides the role of glucosinolate metabolism in response to drought stress[J]. Molecules, 2018, 23(5): 1186. doi: 10.3390/molecules23051186
[97] GOLS R, VAN DAM N M, REICHELT M, et al. Seasonal and herbivore-induced dynamics of foliar glucosinolates in wild cabbage (Brassica oleracea)[J]. Chemoecology, 2018, 28(3): 77-89. doi: 10.1007/s00049-018-0258-4
[98] CARTEA M E, VELASCO P, OBREGÓN S, et al. Seasonal variation in glucosinolate content in Brassica oleracea crops grown in northwestern Spain[J]. Phytochemistry, 2008, 69(2): 403-410. doi: 10.1016/j.phytochem.2007.08.014
[99] VALLEJO F, TOMÁS-BARBERÁN F A, BENAVENTE-GARCÍA A G, et al. Total and individual glucosinolate contents in inflorescences of eight broccoli cultivars grown under various climatic and fertilisation conditions[J]. J Sci Food Agr, 2003, 83(4): 307-313. doi: 10.1002/jsfa.1320
[100] 王辉, 廖永翠, 徐东辉, 等. 普通白菜叶片中硫代葡萄糖苷的季节性变化[J]. 中国蔬菜, 2011, 1(10): 35-40. [101] LEE J H, LEE J, KIM H, et al. Brassinosteroids regulate glucosinolate biosynthesis in Arabidopsis thaliana[J]. Physiol Plant, 2018, 163: 450-458. doi: 10.1111/ppl.2018.163.issue-4
[102] XU L, YANG H, REN L, et al. Jasmonic acid-mediated aliphatic glucosinolate metabolism is involved in clubroot disease development in Brassica napus L[J]. Front Plant Sci, 2018, 9: 750. doi: 10.3389/fpls.2018.00750
[103] YI G E, ROBIN A H, YANG K, et al. Exogenous methyl jasmonate and salicylic acid induce subspecies-specific patterns of glucosinolate accumulation and gene expression in Brassica oleracea L[J]. Molecules, 2016, 21(10): 1417. doi: 10.3390/molecules21101417
[104] THIRUVENGADAM M, BASKAR V, KIM S H, et al. Effects of abscisic acid, jasmonic acid and salicylic acid on the content of phytochemicals and their gene expression profiles and biological activity in turnip (Brassica rapa ssp. rapa)[J]. Plant Growth Regul, 2016, 80(3): 377-390. doi: 10.1007/s10725-016-0178-7
[105] AGUILAR-CAMACHO M, WELTI-CHANES J, JACOBO-VELÁZQUEZ D A. Combined effect of ultrasound treatment and exogenous phytohormones on the accumulation of bioactive compounds in broccoli florets[J]. Ultrason Sonochem, 2019, 50: 289-301. doi: 10.1016/j.ultsonch.2018.09.031



 下载:
下载: