Virulence detection and molecular typing of Staphylococcus aureus isolated from rabbit in Sichuan area
-
摘要:目的
研究四川部分区域兔源金黄色葡萄球菌Staphylococcus aureus的基因型总体结构特征、遗传变异以及毒力因子的分布情况。
方法从四川地区分离41株兔源金黄色葡萄球菌,鉴定femB基因特异性,进行耐甲氧西林金黄色葡萄球菌(Methicillin-resistant S.aureus, MRSA)筛选,并通过PCR法检测13种常见的毒力基因,采用多位点序列分型(Multilocus sequence typing,MLST)和脉冲场凝胶电泳(Pulsed field gel electrophoresis,PFGE)确定基因型特征。
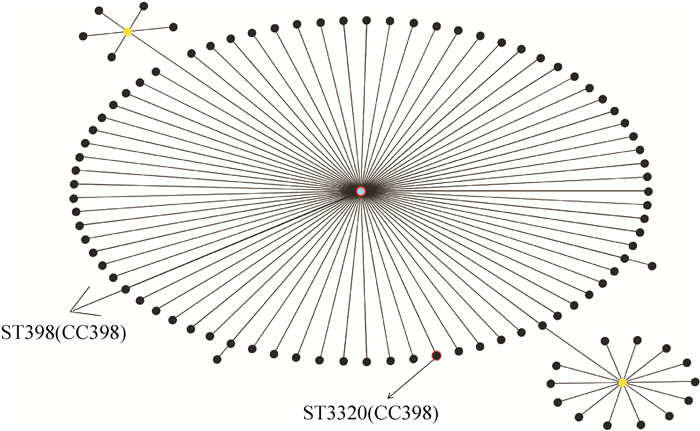
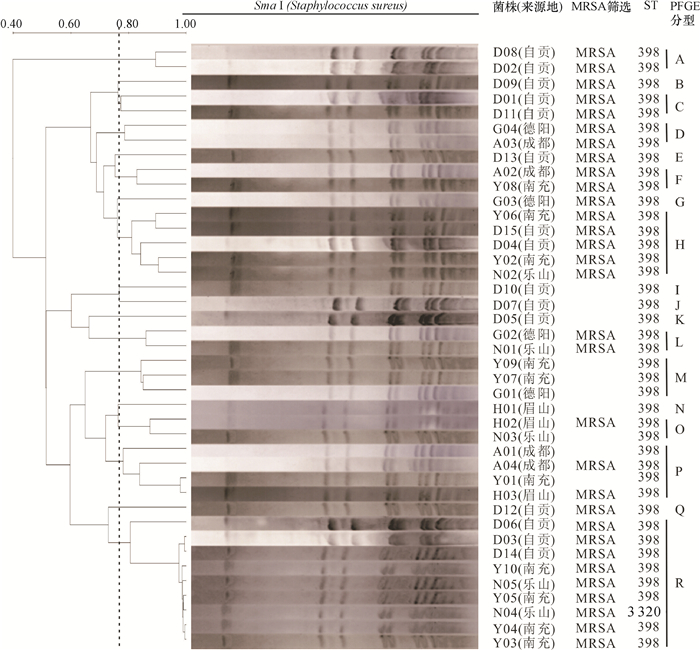
结果41株金黄色葡萄球菌中共检测出MRSA 31株,检出率为75.61%;共检出9种毒力基因,其中nuc、hla、eta和clfA在所有菌株中均存在,而sea、sec、see、hlb和PVL的阳性检出率分别为9.7%、85.4%、80.5%、90.2%和7.3%。MLST分型结果显示,41株金黄色葡萄球菌只存在2种序列型(ST398、ST3320) 和1个克隆群CC398,其中ST398为优势序列型,所占比例为97.6%。PFGE将41株金黄色葡萄球菌分为18个基因型,但不同区域间的基因型条带差异较小。
结论四川调查区域兔源金黄色葡萄球菌毒力因子携带率较高,其对家兔的养殖业存在较大的安全威胁;分型分析说明四川部分区域金黄色葡萄球菌的主要流行菌株遗传变异程度小,菌株间亲缘关系较近。
Abstract:ObjectiveTo understand genotyping characteristics, genetic variation and the distribution of virulence factors of Staphylococcus aureus isolated from rabbits in Sichuan area.
MethodForty-one S. aureus strains were isolated from rabbits in Sichuan area. The strains were identified for femB gene specifity. Methicillin-resistant S. aureus(MRSA) were screened out. Thirteen common virulence genes were detected using PCR. Genotyping characteristics were studied by using multilocus sequence typing (MLST) and pulsed field gel electrophoresis (PFGE).
ResultThere were 31 MRSA strains out of 41 S. aureus strains, and the detection rate was 75.61%. Totally nine virulence genes were detected, nuc, hla, eta and clfA genes existed in all strains, and the positive detection rates of sea, sec, see, hlb and PVL genes were 9.7%, 85.4%, 80.5%, 90.2% and 7.3%, respectively. MLST results showed that 41 S. aureus strains belonged to two sequence types (ST398, ST3320) and one clonal complex CC398. ST398 was the preponderant sequence type, and the proportion was 97.6%. PFGE analysis divided 41 S. aureus strains into 18 banding types, whereas strains from different regions had low variation in banding patterns.
ConclusionS. aureus strains isolated from rabbits in Sichuan area have high carrying rates of virulence factors, and therefore are potential security risk to rabbit farming industry. The genotyping analysis indicates that the prevalent strains of S. aureus isolated from parts of Sichuan Province have low genetic variation, and there are close genetic relationships among different strains.
-
-
表 1 兔源金黄色葡萄球菌毒力基因的检测结果1)
Table 1 PCR detection of virulence genes of Staphylococcus aureus isolated from rabbit
毒力
基因D01~D15 Y01~Y10 N01~N05 A01~A04 G01~G04 H01~H03 MSSA菌株 MRSA菌株 检出率/% 株数 检出率/% 株数 检出率/% 株数 检出率/% 株数 检出率/% 株数 检出率/% 株数 检出率/% 株数 检出率/% 株数 nuc 100.0 15 100.0 10 100.0 5 100.0 4 100.0 4 100.0 3 100.0 10 100.0 31 sea 13.3 2 0 0 40 2 0 0 0 0 0 0 20.0 2 6.5 2 seb 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 sec 93.3 14 100.0 10 100.0 5 50 2 25 1 100.0 3 90.0 9 83.9 26 sed 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 see 100.0 15 80 8 60 3 25 1 75 3 100.0 3 80.0 8 80.6 25 hla 100.0 15 100.0 10 100.0 5 100.0 4 100.0 4 100.0 3 100.0 10 100.0 31 hlb 86.7 13 100.0 10 80 4 75 3 100.0 4 100.0 3 100.0 10 87.1 27 TSST-1 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 PVL 13.3 2 0 0 20 1 0 0 0 0 0 0 20.0 2 3.2 1 eta 100.0 15 100.0 10 100.0 5 100.0 4 100.0 4 100.0 3 100.0 10 100.0 31 etb 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 clfA 100.0 15 100.0 10 100.0 5 100.0 4 100.0 4 100.0 3 100.0 10 100.0 31 1) 检出率:指阳性检出率,株数:指革兰染色阳性的菌株数;菌株D01~D15来源于自贡,Y01~Y10来源于南充,N01~N05来源于乐山,A01~A04来源于成都,G01~G04来源于德阳,H01~H03来源于眉山。 表 2 金黄色葡萄球菌基本信息、等位基因编号及序列号
Table 2 Basic information, allele numbers and sequence types of Staphylococcus aureus strains
菌株 地点 分离
时间等位基因编号 ST arcC aroE glpF gmk pta tpi yqi D01~D15 自贡 2015 3 35 19 2 20 26 39 398 Y01~Y10 南充 2014 3 35 19 2 20 26 39 398 A01~A04 成都 2015 3 35 19 2 20 26 39 398 G01~G04 德阳 2015 3 35 19 2 20 26 39 398 H01~H03 眉山 2014 3 35 19 2 20 26 39 398 N01~N03、N05 乐山 2016 3 35 19 2 20 26 39 398 N04 乐山 2016 3 35 19 249 20 26 39 3 320 -
[1] SCHAUMBURG F, PAULY M, ANOH E, et al. Staphylococcus aureus complex from animals and humans in three remote African regions[J]. Clin Microbiol Infect, 2015, 21(4): 345.e1. doi: 10.1016/j.cmi.2014.12.001
[2] LOWY F D. Staphylococcus aureus infections[J]. New Engl J Med, 1998, 339(27): 2026-2027. doi: 10.1056/NEJM199808203390806?query=prevarrow
[3] REN S Y, GENG Y, WANG K Y, et al. Streptococcus agalactiae infection in domestic rabbits, Oryctolagus cuniculus[J]. Transbound Emerg Dis, 2014, 61(6): e92-e95. doi: 10.1111/tbed.2014.61.issue-6
[4] KRUPA P, BYSTRO AN'U1 J, BANIA J, et al. Genotypes and oxacillin resistance of Staphylococcus aureus from chicken and chicken meat in Poland[J]. Poult Sci, 2014, 93(12): 3179-3186. doi: 10.3382/ps.2014-04321
[5] BREEN J E, HUDSON C D, GREEN M J, et al. Diagnosis and management of intramammary infection caused by Staphyiococcus aureus for dairy cows and herds[J]. Cattle Pract, 2014, 21:189-197.
[6] HIRAMATSU K, HANAKI H, INO T, et al. Methicillin-resistant Staphylococcus aureus clinical strain with reduced vancomycin susceptibility[J]. J Antimicrob Chemoth, 1997, 40(1): 135-136. doi: 10.1093/jac/40.1.135
[7] YOUNGJU J, YOUNSUCK K, HONG S B, et al. Effect of vancomycin plus rifampicin in the treatment of nosocomial methicillin-resistant Staphylococcus aureus pneumonia[J]. Crit Care Med, 2010, 38(1): 175-180. doi: 10.1097/CCM.0b013e3181b9ecea
[8] DZIEKIEWICZMRUGASIEWICZ M. Role of adhesion and biofilm formation by Staphyloccus aureus in bovine mastitis[J]. Med Weter, 2009, 65(2): 84-87. https://www.cabdirect.org/cabdirect/abstract/20093031739
[9] VALSESIA G, ROSSI M, BERTSCHY S, et al. Emergence of SCCmec type IV and SCCmec type V methicillin-resistant Staphylococcus aureus containing the Panton-Valentine leukocidin genes in a large academic teaching hospital in central Switzerland: External invaders or persisting circulators?[J]. J Clin Microbiol, 2010, 48(3): 720-727. doi: 10.1128/JCM.01890-09
[10] MCHUGH E E. MLST analysis of Staphylococcus aureus isolates identified in a pediatric cohort in northwest Houston[D]. Houston: The University of Texas School of Public Health, 2012.
[11] GOLDING G R, CAMPBELL J, SPREITZER D, et al. Pulsed-field gel electrophoresis of Staphylococcus aureus[J]. Methods Mol Biol, 2015, 1301:85-93. doi: 10.1007/978-1-4939-2599-5
[12] O′HARA F P, SUAYA J A, RAY G T, et al. Spa typing and multilocus sequence typing show comparable performance in a macroepidemiologic study of Staphylococcus aureus in the United States[J]. Microb Drug Resist, 2015, 22(1): 88-96. doi: 10.1089/mdr.2014.0238
[13] ALIBAYOV B, BABA-MOUSSA L, SINA H, et al. Staphylococcus aureus mobile genetic elements[J]. Mol Biol Rep, 2014, 41(8): 5005-5018. doi: 10.1007/s11033-014-3367-3
[14] 熊咏民, 莫晓燕, 陈群, 等.聚合酶链反应检测耐甲氧西林金黄色葡萄球菌mecA和femB基因[J].中华微生物学和免疫学杂志, 2003, 23(6): 452. http://www.cnki.com.cn/Article/CJFDTOTAL-DSDX201316032.htm [15] JARRAUD S, MOUGEL C, THIOULOUSE J, et al. Relationships between Staphylococcus aureus genetic background, virulence factors, agr groups (alleles), and human disease[J]. Infect Immun, 2002, 70(2): 631-641. doi: 10.1128/IAI.70.2.631-641.2002
[16] PEACOCK S J, MOORE C E, JUSTICE A, et al. Virulent combinations of adhesin and toxin genes in natural populations of Staphylococcus aureus[J]. Infect Immun, 2002, 70(9): 4987-4996. doi: 10.1128/IAI.70.9.4987-4996.2002
[17] GLASNER C, SABAT A J, DREISBACH A, et al. Rapid and high-resolution distinction of community-acquired and nosocomial Staphylococcus aureus isolates with identical pulsed-field gel electrophoresis patterns and spa types[J]. Int J Med Microbiol, 2013, 303(2): 70-75. doi: 10.1016/j.ijmm.2012.12.005
[18] GOMES-NEVES E, ANTUNES P, TAVARES A, et al. Salmonella cross-contamination in swine abattoirs in Portugal: Carcasses, meat and meat handlers[J]. Int J Food Microbiol, 2012, 157(1): 82-87. doi: 10.1016/j.ijfoodmicro.2012.04.015
[19] 李建平. 奶牛乳房炎金黄色葡萄球菌脉冲场凝胶电泳分型及其主要毒力因子和耐药性的研究[D]. 杭州: 浙江大学, 2009. [20] 陆军. 不同来源耐甲氧西林金黄色葡萄球菌毒力基因的研究[D]. 温州: 温州医学院, 2012. [21] 王英杰. 奶牛乳腺炎金黄色葡萄球菌相关毒力因子的研究[D]. 济南: 山东师范大学, 2008. [22] PRÉVOST G, CRIBIER B, COUPPIÉ P, et al. Panton-Valentine leucocidin and gamma-hemolysin from Staphylococcus aureus ATCC 49775 are encoded by distinct genetic loci and have different biological activities[J]. Infect Immun, 1995, 63(10): 4121-4129. http://iai.asm.org/content/63/10/4121.full.pdf
[23] PANTON P N, VALENTINE F C O. Staphylococcal toxin[J]. Lancet, 1932, 219(5662): 506-508. doi: 10.1016/S0140-6736(01)24468-7
[24] GILLET Y, ISSARTEL B, VANHEMS P, et al. Association between Staphylococcus aureus strains carrying gene for Panton-Valentine leukocidin and highly lethal necrotising pneumonia in young immunocompetent patients[J]. Lancet, 2002, 359(9308): 753-759. doi: 10.1016/S0140-6736(02)07877-7
[25] 童俊, 占志平.金黄色葡萄球菌毒力基因检测及分子分型研究[J].中华微生物学和免疫学杂志, 2015, 35(1):46-50. http://www.cnki.com.cn/Article/CJFDTOTAL-ZSSA201601126.htm [26] WINSTEL V, LIANG C, SANCHEZ-CARBALLO P, et al. Wall teichoic acid structure governs horizontal gene transfer between major bacterial pathogens[J]. Nat Commun, 2013, 4: 2345. http://eprints.gla.ac.uk/85584/1/85584.pdf
[27] XIA G, CORRIGAN R M, WINSTEL V, et al. Wall teichoic acid-dependent adsorption of staphylococcal siphovirus and myovirus[J]. J Bacteriol, 2011, 193(15): 4006-4009. doi: 10.1128/JB.01412-10
[28] VALENTINDOMELIER A S, GIRARD M, BERTRAND X, et al. Methicillin-susceptible ST398 Staphylococcus aureus responsible for bloodstream infections: An emerging human-adapted subclone[J]. PLoS One, 2011, 6(12): e28369. doi: 10.1371/journal.pone.0028369
[29] UHLEMANN A C, PORCELLA S F, TRIVEDI S, et al. Identification of a highly transmissible animal independent Staphylococcus aureus ST398 clone with distinct genomic and cell adhesion properties[J]. Mbio, 2012, 3(2):203-216. http://mbio.asm.org/content/3/2/e00027-12.short?related-urls=yesl3/2/e00027-12
[30] DE NEELING A J, VAN DEN BROEK M J, SPALBURG E C, et al. High prevalence of methicillin resistant Staphylococcus aureus in pigs[J]. Vet Microbiol, 2007, 122(3/4): 366-372. https://www.ncbi.nlm.nih.gov/pubmed/17367960



 下载:
下载: