Study on hydrolysis kinetics of azadirachtin and tructure analysis of hydrolysate
-
摘要:目的
系统研究印楝素在水溶液中的水解。
方法硅胶柱层析法和半制备液相色谱法分离纯化w为44.56%的印楝素原药中的印楝素A,采用核磁共振仪和高效液相色谱定性、定量测定分离得到的印楝素A,建立一种检测水样中印楝素残留的高效液相色谱方法。
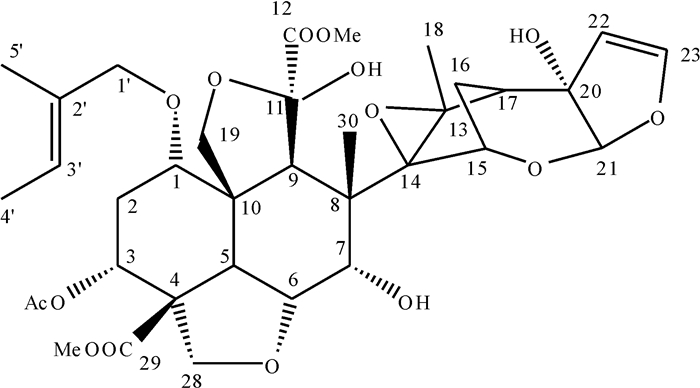
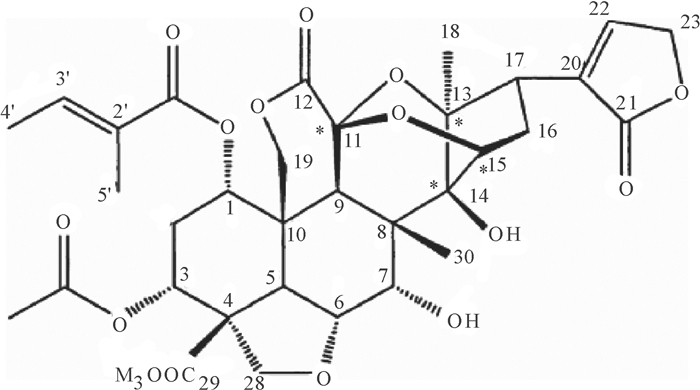
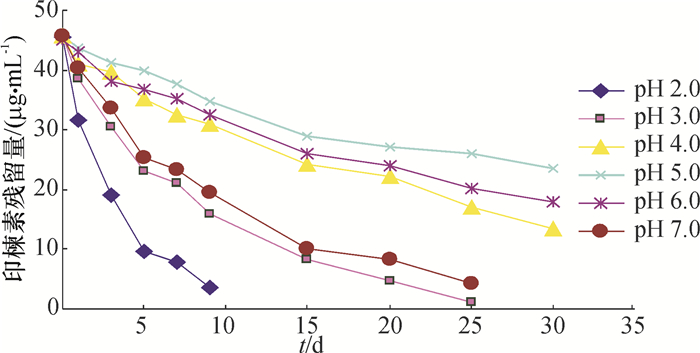
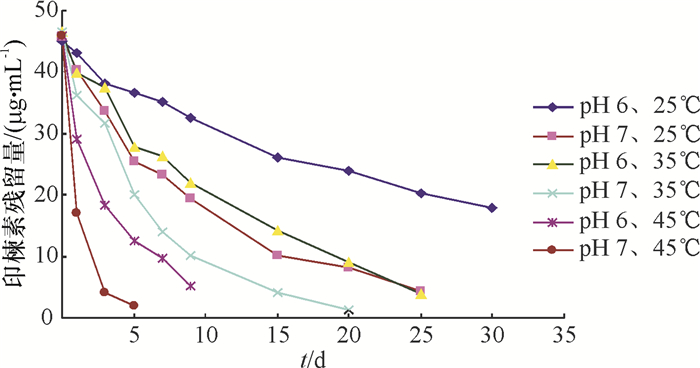
结果核磁共振仪和高效液相色谱测得印楝素A的质量分数分别为90.37%和91.82%。当印楝素添加水平为0.1、1.0和5.0 mg·kg-1时,水样中印楝素的平均回收率为92.53%~94.12%,变异系数为0.35%~0.84%,最小检测质量浓度为0.012 mg·L-1。印楝素在pH 4.0~6.0的缓冲溶液中稳定,当pH大于8.0时,印楝素降解加快,降解半衰期从pH 8.0的14.856 h降到pH 10.0的0.033 h。在pH 6.0的缓冲溶液中,25、35、45 ℃条件下印楝素的降解半衰期分别为24.68、13.69和2.36 d,而在pH 7.0的缓冲溶液中印楝素的降解半衰期分别为9.35、6.51和0.94 d。在pH 2.0的缓冲溶液中分离纯化水解产物得到印楝素A内酯衍生物。
结论印楝素在碱性环境下极不稳定,而在弱酸性环境中比较稳定。温度对印楝素的降解影响很大,随着温度的升高印楝素降解加快。
Abstract:ObjectiveTo study hydrolysis of azadirachtin in water systematically.
MethodAzadirachtin A from the 44.56% azadirachtin TC was isolated and purified by silica column chromatography and semi-preparative high performance liquid chromatography (HPLC). The chemical structure and content of isolated azadirachtin A were identified by nulear magnetic resonance (NMR) and HPLC. A method for determining azaditachtin residue in water by HPLC was established.
ResultThe mass fractions of azadirachtin A were 90.37% and 91.82% detected by NMR and HPLC respectively. When azadirachtin was added with the concentrations of 0.1, 1.0 and 5.0 mg·kg-1, the average recovery rates of azadirachtin from water samples ranged from 92.53% to 94.12%, the variation coefficients ranged from 0.35% to 0.84%, and the minimum detection limit was 0.012 mg·L-1. Azadirachtin was stable in buffer solutions with pH varying from 4.0 to 6.0. When pH was above 8.0, hydrolysis of azadirachtin was accelerated, and the degradation half-life was 14.856 h at pH 8.0 and declined to 0.033 h at pH 10.0. The degration half-lives of azadirachtin in buffer solutions at pH 6.0 were 24.68, 13.69 and 2.36 d under 25, 35 and 45 ℃ temperature respectively, while were 9.35, 6.51 and 0.94 d at pH 7.0. A lactone derivative of azadirachtin was obtained by isolating and purifing hydrolysate in buffer solution at pH 2.0.
ConclusionAzadirachtin is extremely unstable in alkaline environment while relatively stable in weak acid environment. Temperature has a great effect on the degradation of azadirachtin and the degradation accelerates as temperature increases.
-
Keywords:
- azadirachtin /
- residue /
- hydrolysis kinetics /
- hydrolysate /
- silica column chromatography /
- HPLC /
- nuclear magnetic resonance
-
-
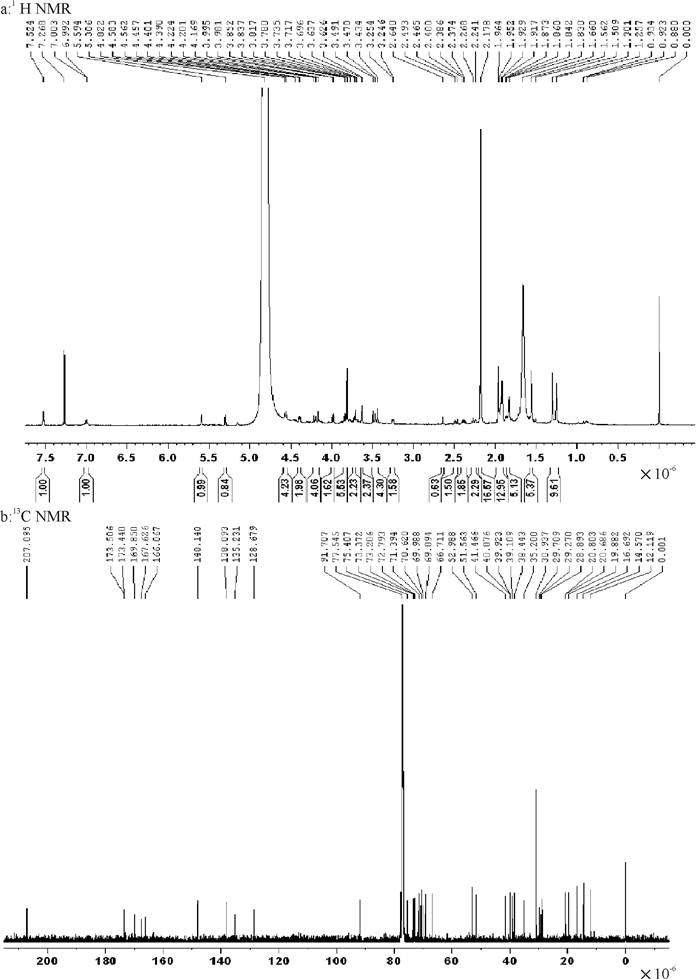
表 1 印楝素的核磁共振碳谱数据13C NMR
Table 1 13C NMR data of azadirachtin ×10-6
碳原子
编号化学位移 文献值[20] 实测值 C-1 70.7 70.6 C-2 29.8 29.7 C-3 67.1 67.0 C-4 45.6 45.6 C-5 37.1 36.9 C-6 74.5 74.3 C-7 76.5 76.5 C-8 50.4 50.3 C-9 44.9 44.7 C-10 52.6 52.5 C-11 104.2 104.2 C-12 169.5 169.8 C-13 70.1 70.1 C-14 68.6 68.8 C-15 73.9 74.0 C-16 25.1 25.0 C-17 48.7 48.7 C-18 20.8 21.0 C-19 69.1 69.0 C-20 83.6 83.4 C-21 107.5 107.6 C-22 108.8 108.7 C-23 147.0 146.8 C-28 73.0 72.9 C-29 166.2 166.3 C-30 18.4 18.5 3-C=O 173.3 173.5 CH3 21.4 21.4 29-OCH3 52.7 52.8 12-OCH3 53.1 53.2 O-Tig; C-1′ 171.8 171.9 C-2′ 128.8 128.6 C-3′ 137.5 137.9 C-4′ 14.2 14.3 C-5′ 11.9 11.9 表 2 印楝素的动力学分析
Table 2 Kinetic analysis of azadirachtin
pH 动力学方程 相关系数(r) 半衰期/d 1.0 Ct=44.31e -0.300 0t 0.999 8 0.10 2.0 Ct=42.57e-0.233 0t 0.997 6 2.98 3.0 Ct=44.61e-0.079 8t 0.997 4 8.68 4.0 Ct=44.03e-0.030 3t 0.994 8 22.87 5.0 Ct=44.85e-0.019 1t 0.991 2 36.33 6.0 Ct=44.36e-0.028 1t 0.997 6 24.68 7.0 Ct=43.04e-0.074 1t 0.997 7 9.35 8.0 Ct=43.73e-0.026 7t 0.975 4 0.62 9.0 Ct=45.16e-0.289 0t 0.998 8 0.10 10.0 Ct=44.99e-0.877 0t 0.999 9 0.03 表 3 印楝素在模拟天然水体不同温度下的降解动力学方程
Table 3 Degradation kinetic equations of azadirachtin in simulated natural water at different temperatures
pH θ/℃ 动力学方程 相关系数(r) 半衰期/d 6.0 25 Ct=44.36e-0.028 1t 0.997 6 24.68 35 Ct=45.08e-0.050 6t 0.994 3 13.69 45 Ct=47.63e-0.294 0t 0.992 4 2.36 7.0 25 Ct=43.04e-0.074 1t 0.997 7 9.35 35 Ct=45.57e-0.106 0t 0.994 5 6.51 45 Ct=45.98e-0.737 0t 0.999 9 0.94 表 4 印楝素在天然水体中的降解动力学方程
Table 4 Degradation kinetic equations of azadirachtin in natural water
水体 动力学方程 相关系数(r) 半衰期/d 珠江水 Ct=46.76e-0.056 9t 0.997 4 12.19 稻田水 Ct=45.98e-0.046 5t 0.989 7 14.91 地表水 Ct=45.72e-0.043 6t 0.996 6 15.90 湖水 Ct=44.79e-0.108 0t 0.994 2 6.43 水库水 Ct=44.96e-0.018 0t 0.995 0 37.06 表 5 印楝素降解半衰期与水体理化性质的相关性分析
Table 5 Analysis of the correlation between the degradation half-life of azadirachtin and physical/chemical properties of water
理化性质 回归方程1) 相关系数(r) pH y=183.570-23.537 0x 0.995 9 总磷含量 y=20.995-11.123 0x 0.285 5 总氮含量 y=27.190-2.367 8x 0.498 0 化学需氧量 y=18.449-0.009 3x 0.044 7 硝酸盐氮含量 y=20.352-2.726 1x 0.376 8 氨氮含量 y=16.873+0.890 6x 0.319 1 1)y为印楝素半衰期,x为对应的理化性质。 -
[1] 荣晓东, 徐汉虹, 赵善欢.植物性杀虫剂印楝的研究进展[J].农药学学报, 2000(2): 9-14. http://www.cnki.com.cn/Article/CJFDTOTAL-NYXB200002001.htm [2] 谭卫红, 宋湛谦.天然植物杀虫剂印楝素的研究进展[J].华南热带农业大学学报, 2004, 10(1): 23-29. http://www.cnki.com.cn/Article/CJFDTOTAL-HEBY200505017.htm [3] 赵淑英, 王秋, 罗万春.印楝植物农药的研究进展[J].济南大学学报, 2004, 18(2): 145-149. http://www.cnki.com.cn/Article/CJFDTOTAL-SDJC200402015.htm [4] 张宗炳, 樊德方, 钱传范, 等.杀虫药剂环境毒理学[M].北京:中国农业出版社, 1989: 25-89. [5] 蔡道基.农药环境毒理学研究[M].北京:中国环境科学出版社, 1999: 78-83. [6] WEI J, FURRER G, SCHULIN R. Kinetics of carbosulfan degradation in the aqueous phase in the presence of a cosolvent[J]. J Environ Qual, 2000, 29(5): 1481-1487. https://www.cabdirect.org/cabdirect/abstract/20003010362
[7] 王连生.有机污染化学[M].北京:科学出版社, 1990: 208-255. [8] SARMAH A K, KOOKANA R S, DUFFY M J, et al. Hydrolysis of triasulfuron, metsulfuron-methyl and chlorsulfuron in alkaline soil and aqueous solutions[J]. Pest Manag Sci, 2000, 56(5): 463-471. doi: 10.1002/(ISSN)1526-4998
[9] WET J, FURRIE G, KAUFMANN S, et al. Influence of clay minerals on the hydrolysis of carbamate pesticides[J]. Environ Sci Technol, 2001, 35(11): 2226-2232. doi: 10.1021/es000179d
[10] 韩丙军, 何书海, 林靖凌, 等.温度影响印楝素A及同系物降解动力学研究[J].现代农药, 2007(6): 30-34. http://www.cnki.com.cn/Article/CJFDTOTAL-NYXD200706012.htm [11] 韩丙军, 陈丽霞, 黄华平, 等.印楝素A降解动力学研究[J].热带作物学报, 2008(1): 97-101. http://www.cnki.com.cn/Article/CJFDTOTAL-RDZX200801023.htm [12] 严亚丽, 陈丽霞, 彭黎旭.水分影响印楝素及同系物降解动力学研究[J].热带农业科学, 2009(5): 30-32. http://www.cnki.com.cn/Article/CJFDTOTAL-RDNK200905007.htm [13] 徐勇, 郭鑫宇, 项盛, 等.植物源杀虫剂印楝素研究开发及应用进展[J].现代农药, 2014(5): 31-37. http://www.cnki.com.cn/Article/CJFDTOTAL-NYXD201405010.htm [14] 罗应兰, 刘宏程.超声波固相萃取-高效液相色谱法同时测定茶叶中印楝素及类似物残留量[J].农药, 2016 (12): 915-917. http://www.cnki.com.cn/Article/CJFDTOTAL-NYZZ201612020.htm [15] 周颖, 安莹, 燕传勇, 等.超高效液相色谱-串联质谱法测定茶叶中印楝素A和B的残留量[J].理化检验, 2016, 52(11): 1292-1296. doi: 10.11973/lhjy-hx201611011 [16] GAI M N, ÁLVAREZ C, VENEGAS R, et al. An HPLC method for determination of azadirachtin residues in bovine muscle[J]. J Chromatogr Sci, 2011, 49(4): 327-331. doi: 10.1093/chrsci/49.4.327
[17] POZO O J, MARIN J M, SANCHO J V, et al. Determination of abamectin and azadirachtin residues in orange samples by liquid chromatography-electrospray tandem mass spectrometry[J]. J Chromatogr A, 2003, 992(1/2): 133-140. http://www.sciencedirect.com/science/article/pii/S002196730300325X
[18] 杨克武, 莫汉宏, 安凤春, 等.有机化合物水解的研究方法[J].环境化学, 1994, 13(3): 206-209. http://www.cnki.com.cn/Article/CJFDTOTAL-HJHX199403004.htm [19] 樊德方.农药残留分析与检测[M].上海:上海科学技术出版社, 1982: 8-26. [20] SHAUN J, DAVID E. Supercritical fluid extraction of oil and triterpenoids from neem seeds[J]. Phytochem Analysis, 1997, 8(5): 228-232. doi: 10.1002/(ISSN)1099-1565



 下载:
下载: